Abstract
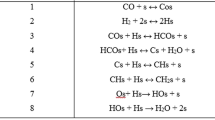
Kinetic models of CO hydrogenation to paraffinic hydrocarbons through Fischer-Tropsch synthesis (FTS) reaction were studied by using Langmuir-Hinshelwood Hougen-Watson (LHHW) model of 16 different reaction steps with a pseudo steady-state assumption (PSSA) on the prototype Pt-promoted Co/Al2O3 catalyst having a granule size of ∼1 mm of spherical γ-Al2O3 support (surface area of 149m2/g). The derived kinetic models from ten sets of experimental data by altering the reaction conditions such as temperatures, pressures, space velocities and H2/CO molar ratios were found to be well fitted with reasonable kinetic parameters and small errors of conversion of CO and hydrocarbon distributions in terms of mean absolute relative residual (MARR) and relative standard deviation error (RSDE). The derived reaction rates and CO activation energy of -86 kJ/mol well correspond to the our previously reported results using power-type catalysts. Based on the LHHW model with PSSA, the possible chemical intermediates on the granule ball-type Co-Pt/Al2O3 surfaces were precisely considered to explain the typical adsorption, initiation, propagation and termination steps of FTS reaction as well as to derive elementary reaction rates with their kinetic parameters and hydrocarbon distributions. The derived kinetic models were further used to verify temperature-profiles in a pilot-scale fixed-bed tubular FTS reactor with a packing depth of 100 cm catalyst, and it confirmed that the temperature gradients were less than 10 °C in a length of reactor by effectively removing the generated heat by an exothermic FTS reaction.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
A. Y. Khodakov, W. Chu and P. Fongarland, Chem. Rev., 107, 1692 (2007).
B. H. Davis, Top. Catal., 32, 143 (2005).
E. Iglesia, Appl. Catal. A, 161, 59 (1997).
M. E. Dry, Catal. Today, 71, 227 (2002).
N. Tsubaki, S. Sun and K. Fujimoto, J. Catal., 199, 236 (2001).
S. H. Song, B. S. Lee, J.W. Bae, P. S. Sai Prasad and K.W. Jun, Catal. Commun., 9, 2282 (2008).
M.H. Woo, J.M. Cho, K.W. Jun, Y. J. Lee and J.W. Bae, Chem-CatChem, 7, 1460 (2015).
S. H. Kwack, M. J. Park, J.W. Bae, S. J. Park, K. S. Ha and K.W. Jun, Fuel Process. Technol., 92, 2264 (2011).
S. J. Park, S. M. Kim, M. H. Woo, J.W. Bae, K.W. Jun and K. S. Ha, Appl. Catal. A: Gen., 419-420, 148 (2012).
B. S. Lee, I. H. Jang, J.W. Bae, S. H. Um, P. J. Yoo, M. J. Park, Y.C. Lee and K.W. Jun, Catal. Surv. Asia., 16, 121 (2012).
S.H. Kwack, J.W. Bae, M. J. Park, S.M. Kim, K.S. Ha and K.W. Jun, Fuel, 90, 1383 (2011).
S.H. Kwack, M. J. Park, J.W. Bae, K. S. Ha and K.W. Jun, Reac. Kinet. Mech. Catal., 104, 483 (2011).
G. Yang, D. Gao, J. Zhang, J. Zhang, Z. Shi, Z. Zhu and D. Xue, RSC Adv., 3, 508 (2013).
C. I. Ahn, H.M. Koo, J.M. Cho, H. S. Roh, J.B. Lee, Y. J. Lee, E. J. Jang and J.W. Bae, Appl. Catal. B: Environ., 180, 139 (2016).
H. Zhu, R. Razzaq, L. Jiang and C. Li, Catal. Commun., 23, 43 (2012).
B.A. Sexton, A. E. Hughes and T.W. Turney, J. Catal., 97, 390 (1986).
R. J. Madon and E. Iglesia, J. Catal., 139, 576 (1993).
A. Lapidus, A. Krylova, V. Kazanskii, V. Borovkov, A. Zaitsev, J. Rathousky, A. Zukal and M. Jancalkova, Appl. Catal. A, 73, 65 (1991).
K. H. Cats and B. M. Weckhuysen, ChemCatChem, 8, 1531 (2016).
C. I. Ahn, H.M. Koo, M. Jin, J.M. Kim, T.G. Kim, Y.W. Suh, K. J. Yoon and J.W. Bae, Micropor. Mesopor. Mater., 188, 196 (2014).
V. Ponec and W.A. van Barneveld, Ind. Eng. Chem. Prod. Res. Dev., 18, 268 (1979).
C.K. Rofer-DePoorter, Chem. Rev., 81, 447 (1981).
K. H. Ernst, E. Schwarz and K. Christmann, J. Chem. Phys., 101, 5388 (1994).
J.T. Kummer, T.W. DeWitt and P. H. Emmett, J. Am. Chem. Soc., 70, 3632 (1948).
E. van Steen and H. Schulz, Appl. Catal. A., 186, 309 (1999).
A.T. Bell, Catal. Rev. Sci. Eng., 23, 203 (1980).
C. S. Kellner and A.T. Bell, J. Catal., 70, 418 (1981).
J. P. Hovi, J. Lahtinen, Z. S. Liu and R. M. Nieminen, J. Chem. Phys., 102, 7674 (1995).
W.A. A. van Barneveld and V. Ponec, J. Catal., 88, 382 (1984).
R.W. Joyner, Catal. Lett., 1, 307 (1988).
H. Schulz, K. Beck and E. Erich, Fuel. Process. Technol., 18, 293 (1988).
C. G. Visconti, E. Tronconi, L. Lietti, R. Zennaro and P. Forzatti, Chem. Eng. Sci., 62, 5338 (2007).
H. J. Jun, M. J. Park, S. C. Baek, J. W. Bae, K. S. Ha and K. W. Jun, J. Natural Gas Chem., 20, 9 (2011).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Koo, H.M., Park, M.J., Moon, D.J. et al. Kinetic models of Fischer-Tropsch synthesis reaction over granule-type Pt-promoted Co/Al2O3 catalyst. Korean J. Chem. Eng. 35, 1263–1273 (2018). https://doi.org/10.1007/s11814-018-0032-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-018-0032-x