Abstract
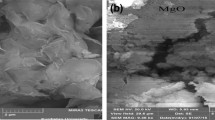
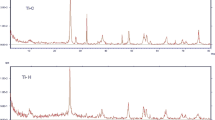
This study considers the feasibility of uptake of cephalexin, an emerging contaminant, from aqueous solutions by using green local montmorillonite (GLM), montmorillonite coated with ZnO (ZnO/GLM) and montmorillonite coated with TiO2 (TiO2/GLM) in the presence of H2O2. Batch adsorption experiments were carried out as a function of pH, initial concentration of the cephalexin, adsorbent dosage, contact time, and temperature. Finally, the adsorbents were characterized by XRD, SEM and FTIR analyses. XRD patterns showed dramatic changes in the adsorbents after loading with the nanoparticles, confirming successful placing of the nanoparticles onto GLM. The GLM mineral surface after nanoparticle loading appears to be fully exposed and more porous with irregular shapes in particles diameters of 1-50 microns. FTIR analyses also confirmed dramatic changes in surface functional groups after modification with these nanoparticles. The results showed that the removal efficiency of cephalexin was better at lower pH values. Totally, the removal efficiency increased with increase in adsorbent dosage and contact time and decreased with concentration and temperature increase. The thermodynamics values of ΔGo and ΔHo revealed that the adsorption process was spontaneous and exothermic. In isotherm study, the maximum adsorption capacities (qm) were obtained to be 7.82, 17.09 and 49.26 mg/g for GLM, ZnO/GLM and TiO2/GLM, respectively. Temkin constant (B T ) showed that adsorption of cephalexin from solution was exothermic for all three adsorbents.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Q. Wu, Z. Li, H. Hong, K. Yin and L. Tie, Appl. Clay Sci., 50(2), 204 (2010).
E. Azizl, M. Fazlzadeh, M. Ghayebzadeh, L. Hemati, M. Beikmohammadi, H.R. Ghaffari, H. R. Zakeri and K. Sharafi, Environ. Protection Eng., 43(1), 183 (2017).
A. J. Watkinson, E. J. Murby and S. D. Costanzo, Water Res., 41(18), 4164 (2007).
H. R. Gaskins, C.T. Collier and D. B. Anderson, Animal Biotechnol., 13(1), 29 (2002).
T. A. Ternes, A. Joss and H. Siegrist, Environ. Sci. Technol., 38(20), 392a (2004).
S. Thiele-Bruhn, J. Plant Nutrition Soil Sci., 166(2), 145 (2003).
M.-S. Miao, Q. Liu, L. Shu, Z. Wang, Y.-Z. Liu and Q. Kong, Process Safety Environ. Protection, 104, Part B, 481 (2016).
Q. Kong, Y.-n. Wang, L. Shu and M.-s. Miao, Desalination Water Treatment, 57(17), 7933 (2016).
M. J. Ahmed and S.K. Theydan, Chem. Eng. J., 211, 200 (2012).
M. Fazlzadeh, K. Rahmani, A. Zarei, H. Abdoallahzadeh, F. Nasiri and R. Khosravi, Adv. Powder Technol., 28(1), 122 (2017).
R. Khosravi, M. Fazlzadehdavil, B. Barikbin and A.A. Taghizadeh, Appl. Surface Sci., 292, 670 (2014).
M. Pirsaheb, A. Dargahi, S. Hazrati and M. Fazlzadehdavil, Desalination Water Treatment, 52(22-24), 4350 (2014).
G. K. Sarma, S. S. Gupta and K. G. Bhattacharyya, J. Environ. Manage., 171, 1 (2016).
M. Fazlzadeh, A. Ahmadfazeli, A. Entezari, A. Shaegi and R. Khosravi, Koomesh, 18(3), 388 (2016).
J. Méndez-Díaz, G. Prados-Joya, J. Rivera-Utrilla, R. Leyva-Ramos, M. Sánchez-Polo, M. Ferro-García and N. Medellín-Castillo, J. Colloid Interface Sci., 345(2), 481 (2010).
H. Abdoallahzadeh, B. Alizadeh, R. Khosravi and M. Fazlzadeh, J. Mazandaran University Medical Sciences, 26(139), 111 (2016).
S. Parastar, S. Nasseri, S. H. Borji, M. Fazlzadeh, A. H. Mahvi, A. H. Javadi and M. Gholami, Desalination Water Treatment, 51(37-39), 7137 (2013).
M. Fazlzadeh, R. Khosravi and A. Zarei, Ecological Engineering, 103, 180 (2017).
M. Leili, M. Fazlzadeh and A. Bhatnagar, Environ. Technol., United Kingdom, 1 (2017).
D. K. Tiwari, J. Behari and P. Sen, World Appl. Sci. J., 3(3), 417 (2008).
M. Fazlzadeh, H. Abdoallahzadeh, R. Khosravi and B. Alizadeh, J. Mazandaran University Medical Sciences, 26(143), 174 (2016).
N. Lian, X. Chang, H. Zheng, S. Wang, Y. Cui and Y. Zhai, Microchim. Acta, 151(1), 81 (2005).
Q. Shi, J. Zhang, C. Zhang, W. Nie, B. Zhang and H. Zhang, J. Colloid Interface Sci., 343(1), 188 (2010).
C. Mena-Duran, M. S. Kou, T. Lopez, J. Azamar-Barrios, D. Aguilar, M. Dominguez, J.A. Odriozola and P. Quintana, Appl. Surface Sci., 253(13), 5762 (2007).
M. Golmohammadi, J. Towfighi, M. Hosseinpour and S. J. Ahmadi, J. Supercritical Fluids, 107, 699 (2016).
Y. Xue, B. Gao, Y. Yao, M. Inyang, M. Zhang, A.R. Zimmerman and K. S. Ro, Chem. Eng. J., 200, 673 (2012).
M. M. Silva, M. M. Oliveira, M. C. Avelino, M. G. Fonseca, R. K. Almeida and E. C. Silva Filho, Chem. Eng. J., 203, 259 (2012).
X. Ren, Z. Zhang, H. Luo, B. Hu, Z. Dang, C. Yang and L. Li, Appl. Clay Sci., 97-98, 17 (2014).
M. Ghaedi, A. G. Nasab, S. Khodadoust, R. Sahraei and A. Daneshfar, J. Ind. Eng. Chem., 21, 986 (2015).
A. B. Albadarin, C. Mangwandi, A.a. H. Al-Muhtaseb, G. M. Walker, S. J. Allen and M. N. M. Ahmad, Chem. Eng. J., 179, 193 (2012).
G. Nazari, H. Abolghasemi and M. Esmaieli, J. Taiwan Inst. Chem. Engineers, 58, 357 (2016).
M. S. Legnoverde, S. Simonetti and E. I. Basaldella, Appl. Surface Sci., 300, 37 (2014).
H. R. Pouretedal and N. Sadegh, J. Water Process Eng., 1, 64 (2014).
M. Ghaedi, M. Ghayedi, S. N. Kokhdan, R. Sahraei and A. Daneshfar, J. Ind. Eng. Chem., 19(4), 1209-17 (2013).
Z. Noorimotlagh, R.D. C. Soltani, G. S. Khorramabadi, H. Godini and M. Almasian, Desalination Water Treatment, 57(4), 1684 (2016).
I. Turku, T. Sainio and E. Paatero, Environ. Chem. Lett., 5(4), 225 (2007).
G. Moussavi, A. Alahabadi, K. Yaghmaeian and M. Eskandari, Chem. Eng. J., 217, 119 (2013).
S. K. Milonjić, J. Serbian Chem. Soc., 72(12), 1363 (2007).
Ş. S. Bayazit and Ö. Kerkez, Chem. Eng. Res. Design, 92(11), 2725 (2014).
O. Pezoti, A. L. Cazetta, I. P. Souza, K. C. Bedin, A. C. Martins, T. L. Silva and V. C. Almeida, J. Ind. Eng. Chem., 20(6), 4401 (2014).
A. C. Martins, O. Pezoti, A. L. Cazetta, K. C. Bedin, D. A. Yamazaki, G. F. Bandoch, T. Asefa, J. V. Visentainer and V. C. Almeida, Chem. Eng. J., 260, 291 (2015).
A. L. Cazetta, A. M. Vargas, E. M. Nogami, M. H. Kunita, M. R. Guilherme, A. C. Martins, T. L. Silva, J. C. Moraes and V. C. Almeida, Chem. Eng. J., 174(1), 117 (2011).
G. Moussavi and R. Khosravi, J. Hazard. Mater., 183(1), 724 (2010).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Khosravi, R., Zarei, A., Heidari, M. et al. Application of ZnO and TiO2 nanoparticles coated onto montmorillonite in the presence of H2O2 for efficient removal of cephalexin from aqueous solutions. Korean J. Chem. Eng. 35, 1000–1008 (2018). https://doi.org/10.1007/s11814-018-0005-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-018-0005-0