Abstract
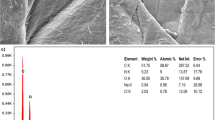
The first application of calcined Cardita bicolor oyster shell (CCBS) for Hg(II) and As(III) adsorption from synthetic and real wastewaters was tested. The main elements in CCBS structure were carbon, oxygen, magnesium, phosphor, and calcium. Effects of different parameters like initial pH, contact time, temperature, and CCBS dosage were assessed. The results showed that the maximum recovery of Hg(II) and As(III) adsorption was determined as C0=10 mg/L, t=80 min, T=25 °C, CCBS dosage=5 g/L, and pH=6 (for mercury ion) and 7 (for arsenic ion). In these conditions, 95.72% Hg(II) and 96.88% As(III) were removed from aqueous solution. The correlation coefficient (R2) values for both adsorbates were obtained >0.98 and >0.96 for Langmuir and Freundlich isotherm models, respectively. Pseudo-second-order kinetic model was more capable to describe kinetic behavior of adsorption process of both metal ions in comparison with pseudo-first-order model. The half life (t1/2) value for Hg(II) and As(III) with initial concentration of 10 mg/L was 4.032 and 4.957 min, respectively. Moreover, thermodynamic parameters of enthalpy (ΔHo), entropy (ΔSo), and Gibbs free energy (ΔGo) were investigated. Two real wastewaters obtained from a leather factory and a landfill leachate were successfully treated using CCBS. The results confirmed that adsorption process of metals ions was exothermic and spontaneous.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
T. Robinson, Waste Manag., 63, 299 (2017).
B. Ramavandi, A. Rahbar and S. Sahebi, Desal. Water Treat., 57, 23814 (2016).
F. Saberzadeh Sarvestani, H. Esmaeili and B. Ramavandi, 3 Biotech, 6, 251 (2016).
A. Rezaee, B. Ramavandi and F. Ganati, Pak. J. Biolog. Sci., 9, 777 (2006).
A. Rezaee, B. Ramavandi, F. Ganati, M. Ansari and A. Solimanian, J. Biolog. Sci., 6, 695 (2006).
K. Johari, N. Saman, S. T. Song, C. S. Chin, H. Kong and H. Mat, Int. Biodeter. Biodegr., 109, 45 (2016).
M. Leili, S. Farjadfard, G. A. Sorial and B. Ramavandi, J. Environ. Manage., 204, 531 (2017).
S.-T. Song, Y.-F. Hau, N. Saman, K. Johari, S.-C. Cheu, H. Kong and H. Mat, J. Environ. Chem. Eng., 4, 1685 (2016).
M. Aryal, M. Ziagova and M. Liakopoulou-Kyriakides, Chem. Eng. J., 162, 178 (2010).
M. Sánchez-Cantú, J. A. Galicia-Aguilar, D. Santamaría-Juárez and L. E. Hernández-Moreno, Appl. Clay Sci., 121, 146 (2016).
A. Guzmán, J. L. Nava, O. Coreño, I. Rodríguez and S. Gutiérrez, Chemosphere, 144, 2113 (2016).
B. Ramavandi, G. Asgari, J. Faradmal, S. Sahebi and B. Roshani, Korean J. Chem. Eng., 31, 2207 (2014).
A. H. Mahvi, M. Shalbafan, R. Nabizadeh, S. Nasseri, S. Jorfi, B. Ramavandi and M. Ahmadi, Toxin Reviews, 36, 52 (2017).
R. Foroutan, M. Madani, M. R. Farani, A. K. Kori, E. Behrad and B. Ramavandi, Int. J. Pharm. Technol., 8, 25133 (2016).
M. Ahmadi, E. Kouhgardi and B. Ramavandi, Korean J. Chem. Eng., 33, 2589 (2016).
M. Fooladvand and B. Ramavandi, Ind. J. Chem. Technol., 22, 183 (2015).
G. Asgari, B. Ramavandi and S. Sahebi, Desal. Water Treat., 52, 7354 (2014).
G. Asgari, B. Ramavandi, L. Rasuli and M. Ahmadi, Desal. Water Treat., 51, 6009 (2013).
W. Peng, H. Li, Y. Liu and S. Song, J. Mol. Liq., 230, 496 (2017).
R. Foroutan, H. Esmaeili, S.D. Rishehri, F. Sadeghzadeh, S. Mirahmadi, M. Kosarifard and B. Ramavandi, Data in Brief, 12, 485 (2017).
S. Delshab, E. Kouhgardi and B. Ramavandi, Data in Brief, 8, 235 (2016).
V. Alipour, S. Nasseri, R. Nabizadeh Nodehi, A. H. Mahvi and A. Rashidi, J. Environ. Health Sci. Eng., 12, 146 (2014).
H.Y. Yen and J.Y. Li, J. Environ. Manage., 161, 344 (2015).
R. Foroutan, F. S. Khoo, B. Ramavandi and S. Abbasi, Desal. Water Treat., 82, 146 (2017).
W. E. Federation and A. P. H. Association, Standard methods for the examination of water and wastewater, American Public Health Association (APHA): Washington, DC, U.S.A. (2005).
D. Alidoust, M. Kawahigashi, S. Yoshizawa, H. Sumida and M. Watanabe, J. Environ. Manage., 150, 103 (2015).
B. Xiang, W. Fan, X. Yi, Z. Wang, F. Gao, Y. Li and H. Gu, Carbohydr. Polym., 136, 30 (2016).
E. Khoramzadeh, B. Nasernejad and R. Halladj, J. Taiwan Ins. Chem. Eng., 44, 266 (2013).
M. Zabihi, A. Haghighi Asl and A. Ahmadpour, J. Hazard. Mater., 174, 251 (2010).
K.D. Brahman, T. G. Kazi, J.A. Baig, H. I. Afridi, S. S. Arain, S. Saraj, M. B. Arain and S. A. Arain, Chemosphere, 150, 320 (2016).
L. Hao, T. Zheng, J. Jiang, G. Zhang and P. Wang, Chem. Eng. J., 292, 163 (2016).
M. S. Podder and C. B. Majumder, J. Mol. Liq., 212, 382 (2015).
M. S. Podder and C. B. Majumder, BioChem. Eng. J., 105, 114 (2016).
M. Ahmadi, H. Rahmani, B. Ramavandi and B. Kakavandi, Desal. Water Treat., 76, 265 (2017).
M. Ahmadi, M. Foladivanda, N. Jaafarzadeh, Z. Ramezani, B. Ramavandi, S. Jorfi and B. Kakavandi, J. Water Supply Res. T., 66, 116 (2017).
M. Shams, I. Nabipour, S. Dobaradaran, B. Ramavandi, M. Qasemi and M. Afsharnia, Fresen. Environ. Bull., 22, 722 (2013).
M. Attari, S. S. Bukhari, H. Kazemian and S. Rohani, J. Environ. Chem. Eng., 5, 391 (2017).
G. Jin, Y. Eom and T. G. Lee, J. Ind. Eng. Chem., 42, 46 (2016).
T.A. Saleh, A. Sarı and M. Tuzen, J. Environ. Chem. Eng., 5, 1079 (2017).
Y. Khambhaty, K. Mody, S. Basha and B. Jha, Sep. Sci. Technol., 43, 1221 (2008).
G. Bayramoğlu, I. Tuzun, G. Celik, M. Yilmaz and M.Y. Arica, Int. J. Min. Process, 81, 35 (2006).
M. Karmacharya, V. K. Gupta, I. Tyagi, S. Agarwal and V. Jha, J. Mol. Liq., 216, 836 (2016).
O.-H. Kwon, J.-O. Kim, D.-W. Cho, R. Kumar, S.H. Baek, M.B. Kurade and B.-H. Jeon, Chemosphere, 160, 126 (2016).
A. Nashine and A. Tembhurkar, J. Environ. Chem. Eng., 4, 3267 (2016).
S. Zavareh, M. Zarei, F. Darvishi and H. Azizi, Chem. Eng. J., 273, 610 (2015).
M.A. Khan, M. Ngabura, T. S.Y. Choong, H. Masood and L.A. Chuah, Biores. Technol., 103, 35 (2012).
C. Xiong, Q. Jia, X. Chen, G. Wang and C. Yao, Ind. Eng. Chem. Res., 52, 4978 (2013).
S.B. Mortazavi, B. Ramavandi and G. Moussavi, Environ. Technol., 32, 251 (2011).
N. Habibi, P. Rouhi and B. Ramavandi, Data in Brief, 13, 749 (2017).
F. Deniz and A. Karabulut, Ecol. Eng., 106, 101 (2017).
Q. Zheng, Z. Li, X. Miao, J. Li, Y. Huang, H. Xia and C. Xiong, Appl. Organomet. Chem., 31, 3546 (2017).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Teimouri, A., Esmaeili, H., Foroutan, R. et al. Adsorptive performance of calcined Cardita bicolor for attenuating Hg(II) and As(III) from synthetic and real wastewaters. Korean J. Chem. Eng. 35, 479–488 (2018). https://doi.org/10.1007/s11814-017-0311-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-017-0311-y