Abstract
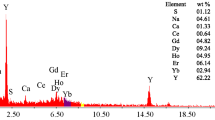
A novel procedure for processing the xenotime mineral concentrate of southwestern Sinai (55.4% RE2O3 and 0.9% U3O8) has been studied. This procedure is based on leaching of uranium and rare earths from the xenotime concentrate using ferric sulfate solution by hydrolytic leaching with ferric ion, wherein ferric iron is precipitated to generate acid for the leaching step. The obtained slurry is washed by water and sulfuric acid. The relevant conditions of the ferric ion leaching of the concentrate and the washing step have been optimized. From the obtained sulfate liquor, rare earths are directly precipitated as oxalates. Uranium is extracted from the REE-free sulfate solution using ion exchange resin Amberlite IRA-400. Highly pure oxide products of the two interesting metal values, REEs and U, have properly been prepared.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
F. Xie, T. Zhang, D. Dreisinger and F. Doyle, Minerals Eng. J., 56, 10 (2014).
A. Jordens, Y.P. Cheng and K.E. Waters, Minerals Eng. J., 41, 97 (2013).
Z. Zhu, Y. Pranolo and Y. C. Cheng, Minerals Eng. J., 77, 185 (2015).
C. K. Gupta and N. Krishnamurthy, Extractive metallurgy of rare earths, CRC Press, Bhabha Atomic Research Centre, Mumbai, India (2004).
M.M. Fawzy, S. M. El Hady, A.R. Bakry and A. S. Al Shami, Egypt J. Appl. Sci., 30(11), 480 (2015).
C.K. Gupta and N. Krishnamurthy, Int. Mater. Rev., 37(1), 197 (1992).
R. Viyayalakshmi, S. L. Mishra, H. Singh and C. K. Gupta, Hydrometallurgy, 61(2), 75 (2001).
P. Alex, A. K. Suri and C.K. Gupta, Hydrometallurgy, 50(3), 331 (1998).
S.M. El Hady, A.R. Bakry, A.S. Al Shami and M.M. Fawzy, Hydrometallurgy, 163, 115 (2016).
F.M. Lever and J. B. Payne, Advances in Extractive Metallurgy, London, the Institution of Mining and Metallurgy, 789 (1968).
R. J. Ring, Hydrometallurgy, 6, 89 (1980).
R. Venter and M. Boylett, Hydrometallurgy Conference 2009, the Southern African Institute of Mining and Metallurgy (2009).
R. J. Bowell, J. Gorgan, M. Hutton-Ashkenny, C. Brough, K. Penman and D. J. Sapsford, Minerals Eng. J., 24(12), 1305 (2011).
A. Costine, A.N. Nikoloski, M.D. Costa, K.F. Chong and R. Hackl, Minerals Eng. J., 53, 84 (2013).
R. Ram, F. A. Charalambous, S. McMaster, M. I. Pownceby, J. Tardio and S. K. Bhargava, Minerals Eng. J., 5051, 83 (2013).
S.K. Bhargava, R. Ram, M. Pownceby, S. Grocott, B. Ring, J. Tardio and L. Jones, Hydrometallurgy, 151, 10 (2015).
P.C. Miller and R.M. Spencer, Pub. No. WO2013173855 A1 (2013).
A. Vuorinen, P. Hiltunen and O. Tuovinen, Hydrometallurgy, 15(3), 297 (1986).
A.D. McKay and Y. Meiitis, AGSO-Geoscience Australia, Mineral Resources Report 1, ISBN 064247161 (2001), (http://www.ga.gov.au/ image_cache/GA9508.pdf).
J. E. Dutrizac, Hydrometallurgy, 73, 11 (2004).
S. I. Abaa, J. Mineralium Deposita, 26(1), 34 (1991).
R. J. Finch and R. C. Ewing, Nuclear Materials J., 190, 133 (1992).
A.M. Dardir, Ph.D. Thesis, Faculty of Science, Ain Shams University, Cairo, Egypt (1997).
J. E. Dutrizac and T.T. Chen, Hydrometallurgy, 98, 128 (2009).
J.A. Sperr, T. N. Solberg and S.W. Becker, Econ. Geol. J., 76(8) (1981).
J. E. Dutrizac and P. A. Riveros, Canadian Institute of Mining, Metallurgy and Petroleum, Montreal, Canada, 967 (2006).
B. Roger and J. Herbert, Clays Clays Minerals J., 45(2), 261 (1997).
Z. Merczenko and M. Balcerzak, Separation, preconcentration and spectrophotometry in inorganic analysis, Elsevier, 341 (2000).
W. Davies and W. Gray, Talanta, 11(8), 1203 (1964).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
El Hady, S.M. A novel procedure for processing of the xenotime mineral concentrate of southwestern Sinai. Korean J. Chem. Eng. 34, 2049–2055 (2017). https://doi.org/10.1007/s11814-017-0095-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-017-0095-0