Abstract
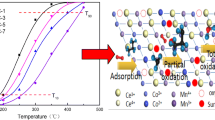
Various physico-chemical techniques and theoretical chemistry computations are used to obtain a deep insight into the mechanism of Ce improving SO2 resistance of the catalyst Mn0.4Ce x /Al2O3 (x stands for the molar ratio of Ce : Al). Theoretical computation with density functional theory (DFT) shows that Ce modification enhances the adsorption energy of SO2 adsorbed on Ce surrounding, resulting in the preferential adsorption of SO2 on Ce surrounding. It protects the surface Mn from SO2 poisoning, leading to a better SO2 resistance. FT-IR and TG results are in good accordance with DFT results. FT-IR results suggest that absorption peaks related to SO4 2− cannot be detected in Mn0.4Ce0.12/Al2O3. Moreover, TG results show that weight loss peaks due to sulfated MnO x decomposition disappears after Ce addition. Therefore, Ce modification inhibits sulfates formation on active components lead to a better resistance to SO2 of Mn0.4Ce0.12/Al2O3.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
T. Boningari, P. R. Ettireddy, A. Somogyvari, Y. Liu, A. Vorontsov, C. A. McDonald and P. G. Smirniotis, J. Catal., 325, 145 (2015).
S. M. Lee, K. H. Park and S. C. Hong, Chem. Eng. J., 195-196, 323 (2012).
Q. Guo, W. Jing, S. Cheng, Z. Huang, D. Sun, Y. Hou and X. Han, Korean J. Chem. Eng., 32, 2257 (2015).
B. Shen, H. Ma and Y. Yao, J. Environ. Sci.-China, 24, 499 (2012).
S. M. Saqer, D. I. Kondarides and X. E. Verykios, Appl. Catal. B Environ., 103, 275 (2011).
F. Cao, J. Xiang, S. Su, P. Wang, S. Hu and L. Sun, Fuel Process. Technol., 135, 66 (2015).
L. Qu, C. Li, G. Zeng, M. Zhang, M. Fu, J. Ma, F. Zhan and D. Luo, Chem. Eng. J., 242, 76 (2014).
M. Wang, H. Liu, Z.-H. Huang and F. Kang, Chem. Eng. J., 256, 101 (2014).
B. Shen, Y. Wang, F. Wang and T. Liu, Chem. Eng. J., 236, 171 (2014).
J. Yu, Z. Si, L. Chen, X. Wu and D. Weng, Appl. Catal. B Environ., 163, 223 (2015).
H. Chang, X. Chen, J. Li, L. Ma, C. Wang, C. Liu, J. W. Schwank and J. Hao, Environ. Sci. Technol., 47, 5294 (2013).
G. S. Qi and R. T. Yang, J. Catal., 217, 434 (2003).
J. Yu, F. Guo, Y. L. Wang, J. H. Zhu, Y. Y. Liu, F. B. Su, S. Q. Gao and G. G. Xu, Appl. Catal. B Environ., 95, 160 (2010).
F. D. Liu and H. He, Catal. Today, 153, 70 (2010).
F. Cao, S. Su, J. Xiang, P. Wang, S. Hu, L. Sun and A. Zhang, Fuel, 139, 232 (2015).
B. X. Shen, X. P. Zhang, H. Q. Ma, Y. Yao and T. Liu, J. Environ. Sci.-China, 25, 791 (2013).
Q. Zhang, Z. Song, P. Ning, X. Liu, H. Li and J. Gu, Catal. Commun., 59, 170 (2015).
Z. Wu, R. Jin, H. Wang and Y. Liu, Catal. Commun., 10, 935 (2009).
R. Jin, Y. Liu, Y. Wang, W. Cen, Z. Wu, H. Wang and X. Weng, Appl. Catal. B Environ., 148, 582 (2014).
P. Yang, X. M. Xue, Z. H. Meng and R. X. Zhou, Chem. Eng. J., 234, 203 (2013).
B. Delley, J. Chem. Phys., 92, 508 (1990).
B. Delley, J. Chem. Phys., 113, 7756 (2000).
J. P. Perdew, K. Burke and M. Ernzerhof, Phys. Rev. Lett., 77, 3865 (1996).
G. Qi and R. T. Yang, J. Phys. Chem. B, 108, 15738 (2004).
W. H. Baur, Acta Crystallogr B, 32, 2200 (1976).
O. T. Sørensen, J. Solid State Chem., 18, 217 (1976).
A. Laachir, V. Perrichon, A. Badri, J. Lamotte, E. Catherine, J. C. Lavalley, J. El Fallah, L. Hilaire, F. Le Normand and E. Quéméré, J. Chem. Soc., Faraday Trans., 87, 1601 (1991).
D. H. Shang, W. Cai, W. Zhao, Y. F. Bu and Q. Zhong, Catal. Lett., 144, 538 (2014).
B. Thirupathi and P. G. Smirniotis, Appl. Catal. B Environ., 110, 195 (2011).
A. Sultana, M. Sasaki and H. Hamada, Catal. Today, 185, 284 (2012).
J. L. Xie, D. Fang, F. He, J. F. Chen, Z. B. Fu and X. L. Chen, Catal. Commun., 28, 77 (2012).
G. Qi and R. T. Yang, J. Phys. Chem. B, 108, 15738 (2004).
D. I. Kondarides and X. E. Verykios, J. Catal., 174, 52 (1998).
S. Yang, W. Zhu, Z. Jiang, Z. Chen and J. Wang, Appl. Surf. Sci., 252, 8499 (2006).
Y. Shao, J. Li, H. Chang, Y. Peng and Y. Deng, Catal. Sci. Technol., 5, 3536 (2015).
K. B. Zhou, X. Wang, X. M. Sun, Q. Peng and Y. D. Li, J. Catal., 229, 206 (2005).
D. A. Tompsett, S. C. Parker and M. S. Islam, J. Am. Chem. Soc., 136, 1418 (2014).
G. A. E. Oxford and A. M. Chaka, J. Phys. Chem. C, 115, 16992 (2011).
Y. Peng, W. W. Yu, W. K. Su, X. Huang and J. H. Li, Catal. Today, 242, Part B, 300 (2015).
Y. Y. Ji, T. Toops and M. Crocker, Catal. Lett., 127, 55 (2009).
Y. Y. Ji, T. J. Toops, J. A. Pihl and M. Crocker, Appl. Catal. B Environ., 91, 329 (2009).
Y. Zhang, X. Zhao, H. Xu, K. Shen, C. Zhou, B. Jin and K. Sun, J. Colloid Interface Sci., 361, 212 (2011).
W. J. Ji, Y. Chen, S. K. Shen, S. B. Li and H. L. Wang, Appl. Surf. Sci., 99, 151 (1996).
H. B. Fu, X. Wang, H. B. Wu, Y. Yin and J. M. Chen, J. Phys. Chem. C, 111, 6077 (2007).
L. Mao, T. Ali, C. Huang and N. Z. Muradov, Int. J. Hydrogen Energy, 36, 5822 (2011).
W. S. Kijlstra, M. Biervliet, E. K. Poels and A. Bliek, Appl. Catal. B Environ., 16, 327 (1998).
W. Xu, H. He and Y. Yu, J. Phys. Chem. C, 113, 4426 (2009).
M. Casapu, O. Kröcher and M. Elsener, Appl. Catal. B Environ., 88, 413 (2009).
Z. P. Zhu, Z. Y. Liu, S. J. Liu, H. X. Niu, T. D. Hu, T. Liu and Y. N. Xie, Appl. Catal. B Environ., 26, 25 (2000).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, X., Li, Z., Zhao, J. et al. Mechanism of Ce promoting SO2 resistance of MnO x /γ-Al2O3: An experimental and DFT study. Korean J. Chem. Eng. 34, 2065–2071 (2017). https://doi.org/10.1007/s11814-017-0092-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-017-0092-3