Abstract
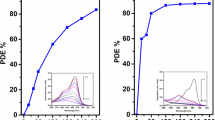
Separation of TiO2 from aqueous suspension is a major constraint in heterogeneous photocatalytic water treatment. As an alternative for existing less effective immobilization techniques, the application of plant based coagulant for the recovery and reuse of TiO2 was investigated for the first time. Aqueous extract derived from seeds of Strychnos potatorum was found to be an effective coagulant for the sedimentation of TiO2. Further, the potential for recovery and reuse of the sedimented photocatalysts TiO2, was investigated by photocatalytic degradation of rhodamine B and terephthalic acid tests. The photocatalytic degradation experiments with recovered catalyst obey pseudo first-order kinetics with enhanced photocatalytic activity than that of the pure TiO2. The investigation of recovered catalysts with XRD, BET, SEM etc., suggests that there is no change in surface and morphological properties when compared with pure TiO2 and the recovered catalysts are highly suitable for recycle and reuse.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
K. Nakata and A. Fujishima, J. Photochem. Photobiol. C, 13, 169, (2012).
C. Pagnout, S. Jomini, M. Dadhwal, C. Caillet, F. Thomas and P. Bauda, Colloids Surf., B, 12, 315, (2012).
A. G. Rincon, C. Pulgarin, N. Adler and P. Peringer, J. Photochem. Photobiol. A: Chem., 139, 233, (2001).
A. R. Khataee, Environ. Technol., 30, 1155, (2009).
P. K. Robertson, J. Robertson and D. W. Bahnemann, J. Hazard. Mater., 211, 161, (2012).
Y. Kikuchi, K. Sunada, T. Iyoda, K. Hashimoto and A. Fujishima, J. Photochem. Photobiol. A, 106, 51, (1997).
D. M. Alrousan, P. S. Dunlop, T. A. McMurray and J. A. Byrne, Water Res., 43, 47, (2009).
A. L. Linsebigler, G. Lu and J. T. Yates, Chem. Rev., 95, 735, (1995).
J. Kim and W. Choi, Energy Environ. Sci., 3, 1042, (2010).
S. Devipriya and S. Yesodharan, Sol. Energy Mater. Sol. Cells, 86, 309, (2005).
H. Sun, Y. Bai, Y. Cheng, W. Jin and N. Xu, Ind. Eng. Chem. Res., 45, 4971, (2006).
S. Kagaya, K. Shimizu, R. Arai and K. Hasegawa, Water Res., 33, 1753, (1999).
A. Fernandez, G. Lassaletta, V. M. Jimenez, A. Justo, A. R. Gonzalez-Elipe, J. M. Herrmann and Y. Ait-Ichou, Appl. Catal. B: Environ., 7, 49, (1995).
A. Rachel, M. Subrahmanyam and P. Boule, Appl. Catal. B: Environ., 37, 301, (2002).
D. Li, S. Zhu, R. H. Pelton and M. Spafford, Colloid. Polym. Sci., 277, 108, (1999).
H. T. Wang, Y. Y. Ye, J. Qi, F. T. Li and Y. L. Tang, Water Sci. Technol., 68, 1137, (2013).
R. Divakaran and V. S. Pillai, Water Res., 38, 2135, (2004).
P. N. Tripathi, N. Chaudhuri and S. D. Bokil, Indian J. Environ. Health, 18, 272, (1976).
S. Biswas, T. Murugesan, K. Maiti, L. Ghosh, M. Pal and B. P. Saha, Phytomedicine, 8, 469, (2001).
M. Adinolfi, M. M. Corsaro, R. Lanzetta, M. Parrilli, G. Folkard, W. Grant and J. Sutherland, Carbohydr. Res., 263, 103, (1994).
J. Qi, Y. Y. Ye, J. J. Wu, H. T. Wang and F. T. Li, Water Sci. Technol., 67, 147, (2013).
APHA, 20th Ed. American Public Health Association, Washington (1998).
Y. Yang, T. Zhang, L. Le, X. Ruan, P. Fang, C. Pan and J. Wei, Sci. Rep., 4, 7045, (2014).
C. Guillard, E. Puzenat, H. Lachheb, A. Houas and J. M. Herrmann, Int. J. Photoenergy, 7, 1, (2005).
W. P. Cheng, Chemosphere, 47, 963, (2002).
J. M. Santos, J. P. Valente, S. M. A. Jorge, P. M. Padilha, M. J. Saeki, G. R. Castro and A. O. Florentino, Orbital: Electron. J. Chem., 5, 233, (2013).
R. A. French, A. R. Jacobson, B. Kim, S. L. Isley, R. L. Penn and P. C. Baveye, Environ. Sci. Technol., 43, 1354, (2009).
M. M. Corsaro, I. Giudicianni, R. Lanzetta, C. E. Marciano, P. Monaco and M. Parrilli, Phytochemistry, 39, 1377, (1995).
J. Yu, H. Yu, B. Cheng and C. Trapalis, J. Mol. Catal. A: Chem., 9, 135, (2006).
H. K. Shon, S. Vigneswaran, I. S. Kim, J. Cho, G. J. Kim, J. B. Kim and J. H. Kim, Environ. Sci. Technol., 41, 1372, (2007).
K. S. Sing, Pure Appl. Chem., 57, 603, (1985).
Y. Wang, L. Zhang, K. Deng, X. Chen and Z. Zou, J. Phys. Chem. C, 111, 2709, (2007).
G. L. Drisko, L. Cao, M. C. Kimling, S. Harrisson, V. Luca and R. A. Caruso, ACS Appl. Mater Interfaces, 1, 2893, (2009).
Y. G. Mishael, P. L. Dubin, R. de Vries and A. B. Kayitmazer, Langmuir, 23, 2510, (2007).
J. F. Porter, Y. G. Li and C. K. Chan, J. Mater. Sci., 34, 1523, (1999).
Author information
Authors and Affiliations
Corresponding author
Additional information
Authors share equal contribution.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Patchaiyappan, A., Saran, S. & Devipriya, S.P. Recovery and reuse of TiO2 photocatalyst from aqueous suspension using plant based coagulant - A green approach. Korean J. Chem. Eng. 33, 2107–2113 (2016). https://doi.org/10.1007/s11814-016-0059-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-016-0059-9