Abstract
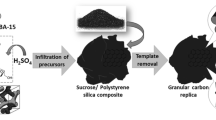
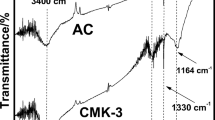
Ordered mesoporous carbon (CMK-3) was synthesized using SBA-15 mesoporous molecular sieve as a template and sucrose as carbon source. The materials were characterized by XRD, TEM and N2 physical adsorption technique. The resulting CMK-3 was used as adsorbent to remove Cd(II) ions from aqueous solution. The effect of pH, contact time and temperature on adsorption process was investigated in batch experiments. The results showed that the removal percentage could reach ca. 90% at the conditions of initial Cd(II) ions concentration of 20 mg/L, dose of 20mg, pH 6.5, contact time of 3h and 293K. Langmuir and Freundlich models were employed to describe the adsorption equilibrium. The kinetics data were described by the pseudo-first-order and pseudo-second-order models, respectively. The adsorption isotherm was well fitted to the Langmuir model, and the adsorption process was well described by the pseudo-second-order kinetic model.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
H. Wang, X. Z. Yuan, Y. Wu, H. J. Huang, G. G. Zeng, Y. Liu, X. L. Wang, N. B. Lin and Y. Qi, Appl. Surf. Sci., 279, 432 (2013).
H. Sukpreabprom, O. Arqueropanyo, W. Naksata, P. Sooksamiti and S. Janhom, Korean J. Chem. Eng., 32, 896 (2015).
S. H. Khorzughy, T. Eslamkish, F. D. Ardejani and M. R. Heydartaemeh, Korean J. Chem. Eng., 32, 88 (2015).
F. Y. Wang, H. Wang and J. W. Ma, J. Hazard. Mater., 177, 300 (2010).
P. Ghosh, A. N. Samanta and S. Ray, Desalination, 266, 213 (2011).
L. H. Liu, J. Wu, X. Li and Y. L. Ling, Sep. Purif. Technol., 103, 92 (2013).
A. Dbrowski, Z. Hubicki, P. Podkocielny and E. Robens, Chemosphere, 56, 91 (2004).
M. Mohsen-Nia, P. Montazeri and H. Modarress, Desalination, 217, 276 (2007).
M. E. Argun, S. Dursun, M. Karatas and M. Guru, Bioresour. Technol., 99, 8691 (2008).
T. Mohammadi, A. Moheb, M. Sadrzadeh and A. Razmi, Sep. Purif. Technol., 41, 73 (2005).
F. P. Zhao, E. Repo, D. L. Yin and M. E. T. Sillanpää, J. Colloid Interface Sci., 409, 174 (2013).
S. P. Chen, J. X. Hong, H. X. Yang and J. Z. Yang, J. Environ. Radioact., 126, 253 (2013).
Z. Huang, X. Zheng, W. Lv, M. Wang, Q. Yang and F. Kang, Langmuir, 27, 7558 (2011).
S. G. Liu, Y. Q. Ding, P. F. Li, K. S. Diao, X. C. Tan, F. H. Lei, Y. H. Zhan, Q. M. Li, B. Huang and Z. Y. Huang, Chem. Eng. J., 248, 135 (2014).
P. Xu, G. M. Zeng, D. L. Huang, C. L. Feng, S. Hu, M. H. Zhao, C. Lai, Z. Wei, C. Huang, G. X. Xie and Z. F. Liu, Sci. Total Environ., 424, 1 (2012).
J. Hu, M. C. Lo and G. H. Chen, Water Sci. Technol., 50, 139 (2004).
G. Zhao, J. Li, X. Ren, C. Chen and X. Wang, Environ. Sci. Technol., 45, 10454 (2011).
V. C. Srivastava, I. D. Mall and I. M. Mishra, Chem. Eng. Process., 47, 1269 (2008).
S. Kumar, V. A. Loganathan, R. B. Gupta and M. O. Barnett, J. Environ. Manage., 92, 2504 (2011).
W. J. Yang, P. Ding, L. Zhou, J. G. Yu, X. Q. Chen and F. P. Jiao, Appl. Surf. Sci., 282, 38 (2013).
M. F. R. Pereira, S. F. Soares, J. J. M. Orfao and J. L. Figuerredo, Carbon, 41, 811 (2003).
V. Gómez, M. S. Larrechi and M. P. Calla, Chemosphere, 69, 1151 (2007).
J. Avom, J. K. Mbadcam, C. Noubactep and P. Germain, Carbon, 35, 365 (1997).
Z. G. Jia, Q. Z. Wang, D. P. Ren and R. S. Zhu, Appl. Surf. Sci., 264, 255 (2013).
R. Ryoo, S. H. Joo and S. Jun, J. Phys. Chem. B, 103, 7743 (1999).
J. Goscianska, A. Olejinik and R. Pietrzak, J. Taiwan Inst. Chem. Eng., 45, 347 (2014).
Y. Tian, P. Liu, X. Wang and H. Lin, Chem. Eng. J., 171, 1263 (2011).
A. Vinu, M. Miyahara, V. Sivamurugan, T. Mori and K. Ariga, J. Mater. Chem., 155, 122 (2005).
D. Y. Zhao, Q. S. Huo, J. L. Feng, B. F. Chmelka and G. D. Stucky, J. Am. Chem. Soc., 120, 6024 (1998).
S. Jun, S. H. Joo, R. Ryoo, M. Kruk, M. Jaroniec, Z. Liu, T. Ohsuna and O. Terasaki, J. Am. Chem. Soc., 122, 10712 (2000).
R. Ahmad, R. Kumar and S. Haseeb, Arabian J. Chem., 5, 353 (2012).
D. Zhao, J. Feng, Q. Huo, N. Melosh, G. H. Fredrickson, B. F. Chmelka and G. D. Stucky, Science, 279, 548 (1998).
L. F. Wang, S. Lin, K. F. Lin, C. Y. Yin, D. S. Liang, Y. Di, P. W. Fan, D. Z. Jiang and F. S. Xiao, Micropor. Mesopor. Mater., 85, 136 (2005).
C. T. Kresge, M. E. Leonowicz, W. J. Roth, J. C. Vartuli and J. S. Beck, Nature, 359, 710 (1992).
H. P. Ye, Q. Zhu and D. Y. Du, Bioresour. Technol., 101, 5175 (2010).
M. J. Baniamerian, S. E. Moradi, A. Noori and H. Salahi, Appl. Surf. Sci., 256, 1347 (2009).
Y. C. Sharma, Uma and S. N. Upadhyay, Energy Fuels, 23, 2983 (2009).
B. Al-Rashdi, C. Tizaoui and N. Hilal, Chem. Eng. J., 183, 294 (2012).
Y. J. Tu, C. F. You and C. K. Chang, J. Hazard. Mater., 235-236, 116 (2012).
P. Xu, G. M. Zeng, D. L. Huang, C. Lai, M. H. Zhao, Z. Wei, N. J. Li, C. Huang and G. X. Xie, Chem. Eng. J., 203, 423 (2012).
M. Doğan and M. Alkan, Chemosphere, 50, 517 (2003).
I. Mobasherpour, E. Salahi and M. Ebrahimi, J. Saudi Chem. Soc., 18, 792 (2014).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lu, L., Zhao, H., Yan, L. et al. Removal characteristics of Cd(II) ions from aqueous solution on ordered mesoporous carbon. Korean J. Chem. Eng. 32, 2161–2167 (2015). https://doi.org/10.1007/s11814-015-0150-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-015-0150-7