Abstract
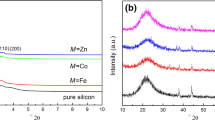
Properties of porous manganese oxide adsorbents for adsorptive removal of tert-butylmercaptan (TBM) from CH4 fuel gas were investigated at ambient temperature and atmospheric pressure. The adsorbents were prepared by oxidation reactions of Mn2+ with KMnO4 and via the sol-gel method by reduction of KMnO4 using fumaric acid as the reducing agent. The effects of preparation method, precursor, temperature, and time for the structure and desulfurization properties of the resulting adsorbents were studied. Cryptomelane octahedral manganese oxide molecular sieve (OMS-2) adsorbents exhibited high breakthrough TBM adsorption (1.3–2.5mmol g-1) with the properties varied by the synthesis condition. The OMS-2-Ac prepared by the oxidation reactions of manganese acetate resulted in smaller OMS-2 crystallites with higher surface area compared to those prepared from manganese sulfate and chloride precursors, and it exhibited an enhanced TBM adsorption uptake. TBM adsorption capacity of OMS-2 could be further enhanced by introducing Cu into the structure. This gave rise to a markedly high TBM breakthrough adsorption (7.4mmol g-1) for Cu-OMS-2 (7.8 wt% Cu doping), which is significantly greater than the values reported for activated carbon, zeolite, and other porous oxide based solid adsorbents at similar conditions in the literature.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
D. Lee, J. Kim, H.C. Lee, K.H. Lee, E.D. Park and H.-C. Woo, J. Phys. Chem. C, 112, 18955 (2008).
S. Satokawa, Y. Kobayashi and H. Fujiki, Appl. Catal. B: Environ., 56, 51 (2005).
P. H. Ho, S.-Y. Lee, D. Lee and H.-C. Woo, Int. J. Hydrogen Energy, 39, 6737 (2014).
C. Song, Catal. Today, 77, 17 (2002).
R. Ferrauto, S. Hwang, L. Shore, W. Ruettinger, J. Lampert, T. Giroux, Y. Liu and O. Ilinich, Annu. Rev. Mater. Res., 33, 1 (2003).
D. D. Whitehurst, T. Isoda and I. Mochida, Adv. Catal., 42, 345 (1998).
H. Cui and S.Q. Turn, Appl. Catal. B: Environ., 88, 25 (2009).
H. Tamai, H. Nagoya and T. Shiono, J. Colloid Interface Sci., 300, 814 (2006).
S. Hernandez, L. Solarino, G. Orsello, N. Russo, D. Fino and G. Saracco, Int. J. Hydrogen Energy, 33, 3209 (2008).
D. Lee, E.-Y. Ko, H. C. Lee, S. Kim and E.D. Park, Appl. Catal. A: Gen., 334, 129 (2008).
Y. H. Kim, H. C. Woo, D. Lee, H. C. Lee and E.D. Park, Korean J. Chem. Eng., 26, 1291 (2010).
C.-L. Hwang and N.-H. Tai, Appl. Catal. B: Environ., 93, 363 (2010).
A. S. H. Salem, Ind. Eng. Chem. Res., 33, 336 (1994).
R.T. Yang, A. J. Hernández-Maldonado and F. H. Yang, Science, 301, 79 (2003).
F.T.T. Ng, A. Rahman, T. Ohasi and M. Jiang, Appl. Catal. B: Environ., 56, 127 (2005).
C. L. Garcia and J. A. Lercher, J. Phys. Chem., 95, 10729 (1991).
S. Velu, X. Ma and C. Song, Ind. Eng. Chem. Res., 42, 5293 (2003).
G. S. Jung, D. H. Park, D. H. Lee, H. C. Lee, S. B. Hong and H. C. Woo, Appl. Catal. B: Environ., 100, 264 (2010).
H.-T. Kim, S.-M. Kim, K.-W. Jun, Y.-S. Yoon and J.-H. Kim, Int. J. Hydrogen Energy, 32, 3603 (2007).
P. Jeevanandam, K. J. Klabunde and S.H. Tetzler, Micropor. Mesopor. Mater., 79, 101 (2005).
P.H. Ho, S.Y. Lee, J. Kim, D. Lee and H.C. Woo, Fuel Process. Technol., 131, 238 (2015).
G. Qiu, H. Huang, S. Dharmarathna, E. Benbow, L. Stafford and S. L. Suib, Chem. Mater., 23, 3892 (2011).
E. K. Nyutu, C.-H. Chen, S. Sithambaram, V.M.B. Crisostomo and S. L. Suib, J. Phys. Chem. C, 112, 6786 (2008).
N. N. Opembe, C. K. King’ondu, A. E. Espinal, C.-H. Chen, E. K. Nyutu, V.M. Crisostomo and S. L. Suib, J. Phys. Chem. C, 114, 14417 (2010).
Q. Feng, H. Kanoh, Y. Miyai and K. Ooi, Chem. Mater., 7, 148, (1995).
H. C. Genuino, S. Dharmarathna, E. C. Njagi, M. C. Mei and S. L. Suib, J. Phys. Chem. C, 116, 12066 (2012).
W. Y. Hernández, M.A. Centeno, S. Ivanova, P. Eloy, E.M. Gaigneaux and J. A. Odriozola, Appl. Catal. B: Environ., 123, 27 (2012).
R. Kumar, S. Sithambaram and S.L. Suib, J. Catal., 262, 304 (2009).
T. Oishi, K. Yamaguchi and N. Mizuno, ACS Catal., 1, 1351 (2011).
O. Ghodbane, F. Ataherian, N.-L. Wu and F. Favier, J. Power Sources, 206, 454 (2012).
C. H. Jiang, S. X. Dou, H. K. Liu, M. Ichihara and H. S. Zhou, J. Power Sources, 172, 410 (2007).
J. Yuan, X. Liu, O. Akbulut, J. Hu, S. L. Suib, J. Kong and F. Stellacci, Nat. Nano., 3, 332 (2008).
S. Ching, J. L. Roark, N. Duan and S. L. Suib, Chem. Mater., 9, 750 (1997).
X.F. Shen, Y.S. Ding, J. Liu, J. Cai, K. Laubernds, R.P. Zerger, A. Vasiliev, M. Aindow and S. L. Suib, Adv. Mater., 17, 805 (2005).
H. Wakita, Y. Tachibana and M. Hosaka, Micropor. Mesopor. Mater., 46, 237 (2001).
J.F. Moulder, W.F. Stickle, P.E. Sobol, K.D. Bomben, J. Chastain and R.C. King, Handbook of X-ray Photoelectron Spectroscopy, Physical Electronic, U.S.A. (1995).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Ho, P.H., Lee, S.C., Kim, J. et al. High performance of manganese oxide octahedral molecular sieve adsorbents for removing sulfur compounds from fuel gas. Korean J. Chem. Eng. 32, 1766–1773 (2015). https://doi.org/10.1007/s11814-015-0031-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-015-0031-0