Abstract
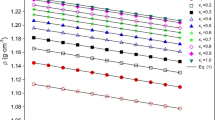
Liquid densities and viscosities are reported for the binary system of 1,2-ethanediamine (EDA)+triethylene glycol (TEG) at T=(298.15, 303.15, 308.15, and 313.15) K. Densities were measured using a capillary pycnometer and viscosities were determined using an Ubbelohde capillary viscometer. The experimental results are compared with data published in the literatures. Based on the density data and kinematic viscosity data, excess molar volumes (V E m ) and deviation in kinematic viscosity (Δν) were calculated and the calculated results were fitted to a Redlich-Kister equation to obtain the coefficients and estimate the standard deviations between the experimental and calculated quantities. The values of V E m are negative in the whole composition range, whereas the values of Δν are positive over the major composition range. From kinematic viscosity data, Gibbs energies of activation of viscous flow (ΔG*), enthalpy of activation for viscous flow (ΔH*), and entropy of activation for the viscous flow (ΔS*) were also calculated.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
P. Luis, T.V. Gerven and B.V. Bruggen, Prog. Energy Combust. Sci., 38, 419 (2012).
J. Koornneef, A. Ramirez and T. Harmelen, Atmos. Environ., 44, 1369 (2010).
C. F. Song and Y. Kitamura, Int. J. Greenh. Gas Con., 7, 107 (2012).
J. Ida and Y. S. Lin, Environ. Sci. Technol., 37, 1999 (2003).
E. Aívarez, F. Cerdeira, D. Gómez-Diaz and J. M. Navaza, J. Chem. Eng. Data, 55, 994 (2010).
B. Han, Y. B. Sun, M. H. Fan and H. S. Cheng, J. Phys. Chem. B, 117, 5971 (2013).
R. Hiwale, S. Hwang and R. Smith, Ind. Eng. Chem. Res., 51, 4328 (2012).
D. Bonenfant, M. Mimeault and R. Hausler, Ind. Eng. Chem. Res., 42, 3179 (2003).
D. Camper, J. E. Bara, D. L. Gin and R. D. Noble, Ind. Eng. Chem. Res., 47, 8496 (2008).
S.Y. Park, K. B. Yi, C.H. Ko, J.-H. Park, J. Kim and W. H. Hong, Energy Fuel, 24, 3704 (2010).
G. N. Patil, P. D. Vaidya and E.Y. Kenig, Ind. Eng. Chem. Res., 51, 1592 (2012).
E. Sada, H. Kumazawa and M. A. Butt, Chem. Eng. J., 13, 213 (1977).
A. Nuchitprasittichai and S. Cremaschi, Int. J. Greenh. Gas Con., 13, 34 (2013).
Z. H. Guo, J. B. Zhang, T. Zhang, C. P. Li, Y. F. Zhang and J. Bai, J. Mol. Liq., 165, 27 (2012).
W. Afzal, A. H. Mohammadi and D. Richon, J. Chem. Eng. Data, 54, 1254 (2009).
A. Valtz, M. Teodorescu, I. Wichterle and D. Richon, Fluid Phase Equilib., 215, 129 (2004).
C. P. Li, J. B. Zhang, T. Zhang, X. H. Wei, E. Q. Zhang, N. Yang, N. N. Zhao, M. Su and H. Zhou, J. Chem. Eng. Data, 55, 4104 (2010).
J. K. Gladden, J. Chem. Eng. Data, 17, 468 (1972).
M. A. Saleh, S. Akhtar and S. Ahmed, J. Mol. Liq., 116, 147 (2005).
J. Ortega, J. Indian Chem. Soc., LXIII, 961 (1986).
B. Vijaya Kumar Naidu, K. Chowdoji Rao and M. C. S. Subha, J. Chem. Eng. Data, 48, 625 (2003).
C. Castellari, J. Chem. Eng. Data, 51, 599 (2006).
A. Kumagai, H. Mochida and S. Takahashi, Int. J. Thermophys., 14, 45 (1993).
N.V. Sastry, R.R. Thakor and M. C. Patel, Int. J. Thermophys., 29, 610 (2008).
N.V. Sastry and M.C. Patel, J. Chem. Eng. Data, 48, 1019 (2003).
F. Han, J. Zhang, G. Chen and X. Wei, J. Chem. Eng. Data, 53, 2598 (2008).
U. R. Kapadi, D.G. Hundiwale, N.B. Patil and M.K. Lande, Fluid Phase Equilib., 25, 267 (2003).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chang, Y., Zhang, J., Li, Q. et al. Excess properties and viscous flow thermodynamics of the binary system 1,2-ethanediamine+triethylene glycol at T=(298.15, 303.15, 308.15, and 313.15) K for CO2 capture. Korean J. Chem. Eng. 31, 2245–2250 (2014). https://doi.org/10.1007/s11814-014-0184-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-014-0184-2