Abstract
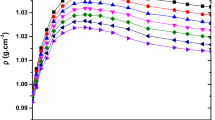
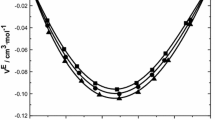
We measured densities (ρ), ultrasonic speeds (u) and viscosities (η) for binary binary mixtures of N-ethylaniline (N-EA) with chlorobenzene (CB), bromobenzene (BB), 1,2-dichlorobenzene (1,2-DCB), 1,3-dichlorobenzene (1,3-DCB), and 1,2,4-trichlorobenzene (1,2,4-TCB) and their pure liquids at 303.15 K and 308.15 K. These experimental data were used to calculate the excess volume (VE), deviations in ultrasonic speeds (Δu), deviation in isentropic compressibility (Δ κ s ), deviation in intermolecular free length (ΔL f ), deviation in acoustic impedance (ΔZ), deviation in viscosity (Δη) and excess Gibbs free energy of activation of viscous flow (G*E). The variations of these properties with composition of binary mixtures suggest loss of dipolar association, difference in size and shape of the component molecules, dipole-dipole interactions and hydrogen bonding between unlike molecules. The viscosity data were correlated with Grunberg and Nissan, Katti and Chaudhri, and Hind et al. equations and the results were compared with the experimental results. The excess parameters were fitted to the Redlich-Kister polynomial equation using multi parametric nonlinear regression analysis to derive the binary coefficients and to estimate the standard deviation.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
M. Gowrisankar, P. Venkateswarlu, K. Siva Kumar and S. Sivarambabu, J. Mol. Liq., 173, 172 (2012).
M. Gowrisankar, P. Venkateswarlu, K. Siva Kumar and S. Sivarambabu, J. Soln., Chem., 42(5), 916 (2013).
M. Gowrisankar, P. Venkateswarlu, K. Siva Kumar and S. Sivarambabu, Korean J. Chem. Eng., 30(5), 1131 (2013).
M. Gowrisankar, P. Venkateswarlu, K. Siva Kumar and S. Sivarambabu, J. Therm. Anal. Calorim., DOI:10.1007/s10973-013-3312z (2013).
V. Syamala, G. Prabhakar, K. Sivakumar and P. venkateswarlu, Phys. Chem. Liq., 44, 127 (2006).
V. Syamala, K. Sivakumar and P. Venkateswarlu, J. Chem. Thermodyn., 38, 1553 (2006).
P. Madhusundhana Reddy, K. Sivakumar and P. Venkatesu, Fluid Phase Equilib., 310, 74 (2011).
J. A. Riddick, W. B. Bunger and T.K. Sakano, Organic solvents physical properties and method of purifications, Wiley Interscience, New York, 2 (1986).
R. Palepu, O. Joan and D. Campell, J. Chem. Eng. Data, 30, 355 (1985).
S. L. Oswal, V. Pandiyan, B. Krishnakumar and P. Vasantharani, Thermochim. Acta, 507, 27 (2010).
G. Chandersekhar, P. Venkatesu and M.V. Prabhakara Rao, Phys. Chem. Liq., 40 181 (2002).
S. C. Bhatia, Ruman Rani, J. Sangwan and Rachna Bhatia, Int. J. Thermophys., 32, 2039 (2011).
A. Ali, A. K. Nain, D. Chand and B. Lal, Indian J. Chem., 44A, 511 (2005).
M. J.V. Rathnam, R. B. Kavitha, R. T. Sayed and M. S. S. Kumar, Eur. Chem. Bull., 2, 434 (2013).
Jaganath and A. P. Dixit, J. Chem. Eng. Data, 29, 313 (1984).
D.V. B. Reddy, K. Ramanjaneyulu and A. Krishnaiah, Phys. Chem. Liq., 27, 221 (1988).
P. Venkateswarlu, N.V. Chowdary and G. K. Raman, Phys. Chem. Liq., 14, 107 (1984).
A. Kumar Nain, J. Mol. Liq., 140, 108 (2008).
F. Kawaizumi, M. Ohno and Y. Miyahara, Bull. Chem. Soc. Jpn., 50, 2229 (1977).
U. Sri Devi, K. Samatha and A. Visvanantasarma, J. Pure Appl. Ultrason., 26, 1 (2004).
R. J. Fort and W. R. Moore, Trans Faraday Soc., 6, 2102 (1965).
R. J. Fort and W. R. Moore, Trans. Faraday Soc., 61, 2102 (1965).
L. Pikkarainan, J. Chem. Eng. Data, 28, 344 (1983).
T. M. Reed and T. E. Taylor, J. Phys. Chem., 63, 58 (1959).
L. Grunberg and A.H. Nissan, Mixture law for viscosity, Nature, 164, 799 (1949).
P. K. Katti and M. H. Chaudhri, J. Chem. Eng. Data, 9, 442 (1964).
R. K. Hind, E. McLaughlin and A. Ubbelohde, Trans Faraday Soc., 56, 328 (1960).
K. C. Kalra, K. C. Singh and D. C. Spah, J. Ind. Chem. Soc., 69, 138 (1992).
O. Redlich and A. T. Kister, Ind. Eng. Chem., 40, 345 (1948).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lakshmi, B.J., Gowrisankar, M., Rambabu, C. et al. Volumetric, ultrasonic and viscometric studies of binary liquid mixures of N-ethylaniline + chlorobenzene, + Bromobeneze, + 1, 2-dichlorobenzene + 1, 3-dichlorobenzene+1, 2, 4-trichlorobenzene at 303.15 and 308.15K. Korean J. Chem. Eng. 31, 881–895 (2014). https://doi.org/10.1007/s11814-013-0235-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-013-0235-0