Abstract
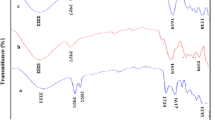
The powdered seeds of Plantago ovata (PSPO) were utilized for the removal of Malachite Green (MG) and Rose Bengal (RB) dyes from aqueous media by batch adsorption. The Fourier transform infra red spectroscopy (FTIR) results showed that both the dyes were adsorbed between the cellulose matrices, and this has been verified from the intensifying and narrowing aromatic C-H bending vibration. The morphology of the dye laden adsorbent was studied by scanning electron microscopy (SEM), which showed that the dyes were adsorbed between the cellulose matrices of the adsorbent. The PSPO was found to be very effective for the removal of MG and RB at pH 7, and equilibrium was attained within 200 min. The kinetic study indicated that the rate limiting step for MG and RB adsorption may be chemisorption and intraparticle diffusion. Adsorption equilibrium data were fitted to Langmuir, Freundlich, Redlich-Peterson and Temkin adsorption isotherms. It is inferred from the equilibrium studies that the adsorption of MG follows the Freundlich isotherm and the adsorption of RB follows the Langmuir isotherm. The maximum monolayer adsorption capacity of the PSPO was found to be 86.23 mg/g for MG and 81.23 mg/g for RB, respectively.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
T. F. Robinson, G. McMullan, R. Marchant and P. Nigam, Bioresour. Technol, 77, 247 (2001).
J. Kent, Van Nostrand Reinhold, New York, N. Y. 7th Ed., 676 (1974).
P. J. Halliday and S. Beszedits, Can. Tex. J, 103, 78 (1986).
G. S. Gupta, G. Prasad and V. N. Singh, Water Res, 24, 45 (1990).
M. Neamtu, A. Yediler, I. Siminiceanu, M. Macoveanu and A. Kellrup, Dyes Pigm, 60, 61 (2004).
I. K. Kapdan and R. Ozturk, J. Hazard. Mater, 123, 217 (2005).
R. K. Wahi, W.W. Yu, Y. P. Liu, M. L. Meija, J.C. Falkner, W. Nolte and V. L. Colvin, J. Molecular Catal. A: Chem, 242, 48 (2005).
V. V. B. Rao and S. R. M. Rao, Chem. Eng. J, 116, 77 (2006).
C. S. Keng, Z. Zainal and A. H. Abdullah, J. Anal. Sci, 12, 451 (2008).
I. Langmuir, J. Ame. Chem. Soc, 40, 1361 (1918).
H.M. F. Freundlich, J. Phys. Chem, 57, 385 (1906).
O. Redlich and D. L. Peterson, J. Phys. Chem, 63, 1024 (1959).
M. J. Temkin and V. Pyzhev, Acta Physicochim. URSS, 12, 217 (1940).
P. K. Baskaran, B. R. Venkatraman and S. Arivoli, E-J. Chem, 8, 9 (2011).
B. H. Hameed and M. I. El-Khaiary, J. Hazard. Mater, 157, 344 (2008).
K. V. Kumar, Dyes Pigm, 74, 595 (2007).
K. V. Kumar and S. Sivanesan, Dyes Pigm, 72, 124 (2007).
G. Annadurai, R. S. Juang and D. J. Lee, J. Hazard. Mater, B92, 263 (2002).
N. Kannan and M.M. Sundaram, Dyes Pigm, 51, 25 (2001).
M. P. Hema and Martin Deva Prasath, J. Sci. Eng, 34, 31 (2009).
F. Banat, S. Al-Asheh and L. Al-Makhadmeh, Process. Biochem, 39, 193 (2003).
Mittal and Alok, J. Hazard. Mater, 133, 196 (2006).
T. Santhi, S. Manonmani, T. Smitha and K. Mahalakshmi, J. Chem, 2, 813 (2009).
I.D. Mall, V. C. Srivastava, N. K. Agarwal and I.M. Mishra, Colloids Surf, 264, 17 (2005).
S. Lagergren, Kungliga Svenska Vetensk Handl, 24, 1 (1898).
Y. S. Ho and G. McKay, Process Biochem, 34, 451 (1999).
W. J. Weber and J.C. Morriss, J. Sanit. Eng. Div. Am. Soc. Civ. Eng, 89, 31 (1963).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Premkumar, M.P., Kumar, V.V., Kumar, P.S. et al. Kinetic and equilibrium studies on the biosorption of textile dyes onto Plantago ovata seeds. Korean J. Chem. Eng. 30, 1248–1256 (2013). https://doi.org/10.1007/s11814-013-0049-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-013-0049-0