Abstract
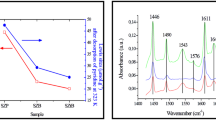
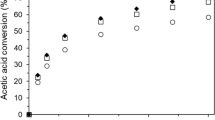
Sulfated zirconia catalysts are proposed for the reversible hydrolysis of ethyl lactate instead of liquid acids. Sulfated zirconia catalysts were prepared by precipitation-impregnation method. The zirconium hydroxide was produced from zirconium oxychloride by adding aqueous ammonia and then impregnated in sulfuric acid. The solid samples were obtained by filtration and evaporation of the mixtures, respectively. After the samples were calcined, the sulfated zirconia catalysts were prepared. The results showed that the catalyst prepared by evaporation has higher catalytic activity. The physicochemical characteristics of the sulfated zirconia catalysts were studied by thermal analysis, X-ray powder diffraction (XRD), temperature programmed desorption of ammonia (NH3-TPD) and N2 adsorption-desorption, respectively. By the precipitation-impregnation-evaporation method, the optimal sulfated zirconia catalyst of tetragonal phase was prepared under liquid-solid ratio of 5ml/g, 1 mol/L of H2SO4 and calcination at 650 °C for 3 h. The conversion of the ethyl lactate was 87.8% in 3 h at 85 °C with the catalyst loading 2 wt% and initial molar ratio of water to ethyl lactate 20: 1.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Y. J. Wee, J. N. Kim and H.W. Ryu, Food Technol. Biotechnol., 44, 163 (2006).
M. T. Sanz, R. Murga, S. Beltran, J. L. Cabezas and J. Coca, Ind. Eng. Chem. Res., 43, 2049 (2004).
R.L. Martins and M. Schmal, Appl. Catal. A: Gen., 308, 143 (2006).
T. A. Peters, N. E. Benes and J.T. F. Keurentjes, Appl. Catal. A: Gen., 317, 113 (2007).
A. Izci and F. Bodur, React. Funct. Polym., 67, 1458 (2007).
T. Peters, N. Benes, A. Holmen and J. Keurentjes, Appl. Catal. A: Gen., 297, 182 (2006).
X. H. Sun, Q. H. Wang, W. C. Zhao, H. Z. Ma and K. Sakata, Sep. Purif. Technol., 49, 43 (2006).
K. Suwannakarn, E. Lotero, J.G. Goodwin Jr. and C. Lu, J. Catal., 255, 279 (2008).
N. Katada, T. Tsubaki and M. Niwa, Appl. Catal. A: Gen., 340, 76 (2008).
X. H. Qi, M. Watanabe, T. M. Aida and R.L. Smith Jr., Catal. Commun., 10, 1771 (2009).
N. Lohitharn and Jr. J. G. Goodwin, J. Catal., 245, 198 (2007).
J. Deutsch, H. A. Prescott, D. Müller, E. Kemnitz and H. Lieske, J. Catal., 231, 269 (2005).
G.D. Yadav and P.H. Mehta, Ind. Eng. Chem. Res., 33, 2198 (1994).
S. Saravanamurugan and A. Riisager, Catal. Commun., 17, 71 (2012).
W. X. Li, Y. X. Ni and W. H. Xing, Adv. Mater. Res., 233, 1529 (2011).
H. Matsuhashi, H. Nakamura, T. Ishihara, S. Iwamoto, Y. Kamiya, J. Kobayashi, Y. Kubota, T. Yamada, T. Matsuda, K. Matsushita, K. Nakai, H. Nishiquchi, M. Oqura, N. Okazaki, S. Sato, K. Shimizu, T. Shishido, S. Yamazoe, T. Takequchi, K. Tomishiqe, H. Yamashita, M. Niwa and N. Katada, Appl. Catal. A: Gen., 360, 89 (2009).
C. Morterra, G. Cerrato and M. Signoretto, Catal. Lett., 41, 101 (1996).
D. H. Guan, M. Q. Fan, J. Wang, J. Wang, Y. Zhang, Q. Liu and X.Y. Jing, Mater. Chem. Phys., 122, 278 (2010).
B.M. Reddy, P.M. Sreekanth and P. L. Ataullah, J. Mol. Catal. A: Chem., 244, 1 (2006).
K. Arata and M. Hino, Mater. Chem. Phys., 26, 213 (1990).
R. Srinivasan and B. H. Davis, Prepr. Am. Chem. Soc. Div. Petrol. Chem., 36, 635 (1991).
D. Farcasiu, J.Q. Li and S. Cameron, Appl. Catal. A: Gen., 154, 173 (1997).
Z. J. Kang, H. Z. Ma and B. Wang, Ind. Eng. Chem. Res., 48, 9346 (2009).
S. R. Kirumakki, N. Nagaraju and K.V. R. Chary, Appl. Catal. A: Gen., 299, 185 (2006).
A. I. Ahmed, S. A. El-Hakam, S. E. Samra, A. A. EL-Khouly and A. S. Khder, Colloids Surf. A: Physicochem. Eng. Aspects, 317, 62 (2008).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, W., Ni, Y., Liu, W. et al. Preparation and properties of sulfated zirconia for hydrolysis of ethyl lactate. Korean J. Chem. Eng. 30, 1222–1228 (2013). https://doi.org/10.1007/s11814-013-0036-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-013-0036-5