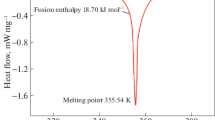
Abstract
Triclocarban solubility in six pure alcohols was determined in the temperature interval from 278.15 to 318.15 K. The experimental solubility data were correlated by the Wilson, the nonrandom two liquid (NRTL) and the universal quasi-chemical (UNIQUAC) models. The data are well fitted with all three models for the six pure alcohols studied here. Also, ab initio geometry optimization of triclocarban was performed using the density functional theory (DFT) based on DMol3 method.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
D. L. Breneman, J.M. Hanifin, C. A. Berge, B.H. Kewick and P.B. Neumann, Cutaneous Medicine for the Practitioner, 66, 296 (2000).
S. Luby, M. Agboatwalla, D. Feikin, J. Painter, W. Billhimer, A. Altaf and R. Hoekstra, The Lancet, 366, 225 (2005).
O. Exner and H. M. Hoffmann, US Patent, 6,121,214 (2000).
L.R. Charlton and J.T. McGillycuddy, US Patent, 6,224,886 (2001).
A. C. Moffat, M. D. Osselton and B. Widdop, Clarke’s Analysis of Drugs and Poisons, 3rd Ed., Vol. 2, Pharmaceutical Press, London (2004).
D. M. Arogón, A. Sosnik and F. Martínez, J. Sol. Chem., 38, 1493 (2009).
Q. S. Ki, Z. Ki and S. Wang, J. Chem. Eng. Data, 52, 151 (2007).
D. Wei and L. Chen, Fluid Phase Equilib., 277, 9 (2009).
R. R. Fu, W. D. Yan and M. Zhu, J. Chem. Eng. Data, 49, 262 (2004).
G.M. Wilson, J. Am. Chem. Soc., 86, 127 (1964).
H. Renon and J. M. Prausnitz, AIChE J., 14, 135 (1968).
D. S. Abrams and J. M. Prausnitz, AIChE J., 21, 116 (1975).
B. N. Taylor and C. E. Kuyatt, Guidelines for Evaluating and Expressing the Uncertainty of NIST Measurement Results, 1994 Ed., NIST.
J. B. Dalton and C. L. A. Schmidt, J. Biol. Chem., 103, 549 (1933).
DIPPR 801 Database, Design Institute for Physical Property Data, American Institute of Chemical Engineers.
B. E. Polling, J. M. Prausnitz and J. P. O’Connell, The Properties of Gases and Liquids, 5th Ed., McGrow-Hill, New York (2001).
L. Constantinou, R. Gani and J. P. O’Connell, Fluid Phase Equilib., 103, 11 (1995).
S. I. Sandler, Chemical, Biochemical, and Engineering Thermodynamics, 4th Ed., John Wiley & Sons, Inc., New Jersey (2005).
J.M. Prausnitz, R.N. Lichtenthaler and E.G. de Azevedo, Molecular Thermodynamics of Fluid-Phase Equilibria, 3rd Ed., Prentice Hall PTR, New Jersey (1999).
D. M. Aragón, M. A. Ruidiaz, E. F. Vargas, C. Bregni, D. A. Chiappetta, A. Sosnik and F. Martínez, J. Chem. Eng. Data, 53, 2576 (2007).
B. Delley, J. Chem. Phys., 113, 7756 (2000).
J. P. Perdew, K. Burke and M. Ernzerhof, Phys. Rev. Lett., 77, 3865 (1996).
J. Chang, Korean Chem. Eng. Res., 49, 361 (2011).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Lim, J., Jang, S., Kim, H. et al. Solubility of triclocarban in pure alkanols at different temperatures. Korean J. Chem. Eng. 30, 181–186 (2013). https://doi.org/10.1007/s11814-012-0099-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-012-0099-8