Abstract
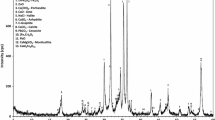
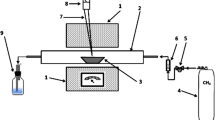
The paper deals with the extraction of copper from the deposited material of the liner of the electrostatic precipitator (ESP) of the copper smelter plant. These precipitates of ESP liner (ESP dust) generally contain mixed phases of copper and arsenic. An attempt is made to extract copper from ESP dust, subsequently removing arsenic from the leach liquor. The ESP dust containing paramelaconite (6CuO·Cu2O), α-domeykite (Cu3As), metadomeykite (Cu3As), enargite (Cu3AsS4) and (Cu,Fe) SO4·H2O is not a naturally occurring geological mineral; thus comparatively high acid concentration and temperature are required to break the matrix of this mixed material so as to liberate the content. The leaching efficiency of copper was 97% at 97 °C. The acid concentration of 1.5M and pulp density of 20% was found to be optimum. The removal of arsenic as ferric arsenate was carried out in two stages: increasing the pH and precipitation of arsenic by adjusting pH of the solution and by adding ferrous sulfate and hydrogen peroxide. The optimum removal of arsenic was 95% when pH was raised to 2.35 followed by precipitation. The key progression of the process is the recovery of copper from ESP dust as well as removal of arsenic from the leach liquor.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
P. D. Oudenne and F. A. Olson, Metall Trans., 14B, 33 (1983).
A. Künkül, M.M. Kocakerim and S. A. Yapici, Int. J. Miner. Process., 41, 167 (1994).
A. Ekmekyapar, R. Oya and A. Künkül, Chem. Biochem. Eng. Q., 17, 261 (2003).
O. N. Ata, S. Çolak, Z. Ekinci and M. Çopur, Chem. Eng. Technol., 24, 409 (2001).
D. Bingol and M. Canbazoglu, Hydrometallurgy, 72, 159 (2004).
K. S. Rao, K. L. Narayana, K.M. Swamy and J. S. Murthy, Metall Mater Trans., 28B, 721 (1997).
A. I. Orlov, Izv. Vyssh. Uchebn. Zaved. Tsventn. Metall., 1, 85 (1975) (in Russian) (Chemical abstract).
P. Priest, L. J. Pauwels and J. Naud, J. Inorg. Nuclear. Chem., 36, 2493 (1974).
J. Viñals, A. Roca, M.C. Hernández and O. Benavente, Hydrometallurgy, 68, 183 (2003).
O. Herreros, R. Quiroz, M. C. Hernández and J. Viñals, Hydrometallurgy, 64, 153 (2002).
R. Padilla, G. Rodríguez and M. C. Ruiz, Hydrometallurgy, 100, 152 (2010).
W. Tongamp, Y. Takasaki and A. Shibayama, Hydrometallurgy, 98, 213 (2009).
A. Janin, P. Drogui, J. F. Blais and G. Mercier, J. Hazard. Mater., 96, 318 (2009).
V. Chandra, J. Park, Y. Chun, J.W. Lee, I. C. Hwang and K. S. Kim, ACS Nano, 4, 3979 (2010).
S. R. Chowdhury and E. K. Yanful, Water Environ. J., 25, 429 (2011).
J. Y. Park and J. H. Kim, J. Nano Particle Res., 13, 887 (2011).
M. J. Kim, Geo Chem.: Exploration, Environment, Analysis, 10, 407 (2010).
S. Singhania, Q. Wang, D. Filippou and G. P. Demopoulos, Metall Mater Trans., 36B, 327 (2005).
S. Singhania, Q. Wang, D. Filippou and G. P. Demopoulos, Metall Mater Trans. B, 37, 189 (2006).
C. H. Mary, E. S. Madeline, R. J. Donald and M.G. Martha, Environ. Sci. Technol., 40, 6709 (2006).
M. C. Bluteau and G. P. Demopoulos, Hydrometallurgy, 87, 163 (2007).
T. Fujita, R. Taguchi, M. Abumiya, M. Matsumoto, E. Shibata and T. Nakamura, Hydrometallurgy, 90, 85, (2008).
T. Fujita, R. Taguchi, M. Abumiya, M. Matsumoto, E. Shibata and T. Nakamura, Hydrometallurgy, 90, 92 (2008).
T. Fujita, R. Taguchi, E. Shibata and T. Nakamura, Hydrometallurgy, 96, 300 (2009).
D. E. Voigt, S. L. Brantley and R. J.C. Hennet, Appl. Geo. Chem., 11, 633 (1996).
L. Yang, R. J. Donahoe and J. C. Redwine, Sci. Total Environ., 387, 28 (2007).
J. Schaller, M. Mkandawire and E.G. Dudel, Environ. Pollut., 158, 2454 (2010).
J. Viñals, A. Sunyer, P. Molera, M. Cruells and N. Llorca, Hydrometallurgy, 104, 247 (2010).
A. Morales, M. Cruells, A. Roca and R. Berg, Hydrometallurgy, 105, 148 (2010).
A. Shibayama, Y. Takasaki, T. William, A. Yamatodani, Y. Higuchi, S. Sunagawa and E. Ono, J. Hazard. Mater., 181, 1016 (2010).
Z. F. Xu, Q. Li and H. P. Nie, Trans. Nonfer. Met. Soc. China, 20, s176 (2010).
D. Paktunc, J. Dutrizac and V. Gertsman, Geochim. Cosmochim. Acta, 72, 2649 (2008).
D. Langmuir, J. Mahoney and J. Rowson, Geochim. Cosmochim. Acta, 70, 2942 (2006).
D. Paktunc and K. Bruggeman, Appl. Geochem., 25, 674 (2010).
A. I. Vogel, A text book of inorganic analysis, 3rd Ed., The English Language Book Society and Longmans, Green and Co., LTD., London, 358 (1961).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sahu, N.K., Dash, B., Sahu, S. et al. Extraction of copper by leaching of electrostatic precipitator dust and two step removal of arsenic from the leach liquor. Korean J. Chem. Eng. 29, 1638–1642 (2012). https://doi.org/10.1007/s11814-012-0081-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-012-0081-5