Abstract
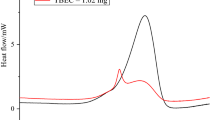
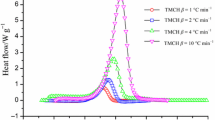
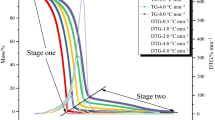
Tert-butyl(2-ethylhexyl)monoperoxy carbonate (TBEHC) 95 mass% is intrinsically a very unstable substance that can induce self-decomposition even under normal atmospheric condition. During storage, TBEHC 95mass% can release an enormous amount of heat if the temperature is higher than the recommended storage temperature, due to the self-accelerating reaction having been ignited. In this study, TBEHC 95mass% was tested by differential scanning calorimetry (DSC) under five heating rates (1, 2, 4, 6, and 8 °C/min) and four isothermal conditions (120, 125, 130, and 135 °C) to evaluate the basic kinetic and safety parameters of time to maximum rate (TMR), self-accelerating decomposition temperature (SADT), and temperature of no return (TNR). Under runaway reaction TBEHC 95 mass% releases a great quantity of heat. This study establishes an important guiding principle for related manufacturing processes worldwide.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
J.M. Tseng, C. M. Shu, J. P. Gupta and Y. F. Lin, Ind. Eng. Chem. Res., 46, 8738 (2007).
X. R. Li, J. H. Sun, H. Koseki and K. Hasegawa, J. Hazard. Mater., A120, 51 (2005).
F. Sevim, F. Demir M. Bilen and H. Okur, Korean J. Chem Eng., 23, 736 (2006).
B. Ning, R. Hu, H. Zhang, Z. Xia, P. Guo, R. Liu, G. Lu and J. Jiang, Thermochim. Acta, 416, 47 (2004).
Z. Hai, X. Zhiming, G. Pengjiang, H. Rongzu, G. Shengli, N. Binke, F. Yan, S. Qizhen and L. Rong, J. Hazard. Mater., 94, 205 (2002).
J.M. Tseng, C.M. Shu and Y.C. Yu, Korean J. Chem. Eng., 22, 797 (2005).
J. H. Cha, K. S. Kim, S. Choi, S. H. Yeon, H. Lee, H. S. Kim and H. Kim, Korean J. Chem. Eng., 22, 945 (2005).
P. Y. Yeh, C.M. Shu and Y. S. Duh, Ind. Eng. Chem. Res., 42, 1 (2003).
R. H. Chang, J.M. Tseng, J.M. Jehng, C.M. Shu and H.Y. Hou, J. Therm. Anal. Cal., 83, 57 (2006).
Y.W. Wang, C.M. Shu, Y. S. Duh and C. S. Kao, Ind. Eng. Chem. Res., 40, 1125 (2001).
H.Y. Hou, T. S. Liao, Y. S. Duh and C. M. Shu, J. Therm. Anal. Cal., 83, 167 (2006).
Y. S. Duh, C. C. Hsu, C. S. Kao and S.W. Yu, Thermochim. Acta, 285, 67 (1996).
H. J. Liaw, C. J. Chen and C. C. Yur, J. Loss Prev. Process Ind., 14, 371 (2001).
C. K. Cho, K. S. Chang and T. S. Cale, Korean J. Chem. Eng., 10, 195 (1993).
K. S. Kim and J.W. Ko, Korean J. Chem. Eng., 22, 26 (2005).
K. S. Park and J. Y. Kim, Korean J. Chem. Eng., 18, 285 (2001).
C. Oommen and S. R. Jain, J. Hazard. Mater., A67, 253 (1999).
H. E. Kissinger, Anal. Chem., 29, 1702 (1957).
T. Ozawa, Thermochim. Acta, 203, 159 (1992).
H. G. Fisher and D. D. Goetz, J. Loss Prev. Process. Ind., 6, 183 (1993).
A. A. Kossoy and Y.G. Akhmetshin, Process Saf. Prog., 26(3), 209 (2007).
Safe storage and handling of reactive materials, Published by Center for Chemical Process Safety of the AIChE, New York, USA, 226 (1995).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, LY., Su, CH., Chuang, KY. et al. Isothermal and non-isothermal kinetic and safety parameter evaluation of tert-butyl(2-ethylhexyl)monoperoxy carbonate by differential scanning calorimetry. Korean J. Chem. Eng. 29, 1292–1297 (2012). https://doi.org/10.1007/s11814-012-0001-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-012-0001-8