Abstract
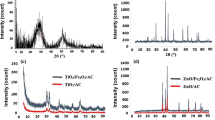
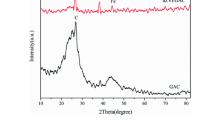
Activated Carbon supported Fe(0) nanoparticles (AC-Fe(0)) were applied to the reductive removal of nitrate to investigate the effects of AC support on the reactivity of Fe(0) nanoparticle. XRD, SEM and EDS, XPS analyses on AC-Fe(0) revealed that AC-Fe(0) is more susceptible to oxidation compared to the unsupported Fe(0) nanoparticles, and that the extent of oxidation of the AC-Fe(0) particles will vary depending on the ratios of AC to Fe(0). Nitrate reduction rate of AC-Fe(0) was much slower than that of unsupported Fe(0) nanoparticles. AC-Fe(0) (0.5: 1) particles reduced the nitrate to ca. 40% of the initial concentration, and AC-Fe(0) (5: 1) particles performed poorly with only 10% removal of the nitrate. Besides the deactivation of AC-Fe(0) due to corrosion of Fe(0), the mass transport limitation caused by the thick layering of Fe(0) on porous AC seemed to be another negative factor for the decreased reactivity of AC-Fe(0).
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
J. Kroschwitz and M. How-Grant, Eds. Encyclopedia of chemical technology, 4th Ed., J. Wiley & Sons, New York, 5. 340 (1993).
J.-J. Li, X.-Y. Xu, Z. Jiang, Z.-P. Hao and C. Hu, Environ. Sci. Technol., 39, 1319 (2005).
M. Cho, E. Kim, K.-H. Lee and S. Ahn, J. Environ. Sci., 17, 711 (2008).
S. M. Ponder, J.G. Darab and T. E. Mallouk,. Environ. Sci. Technol., 34, 2564 (2000).
M. S. Ponder and T. E. Mallouk, U. S. Patent, 6,689, 485 B2 (2004).
J.G. Darab, A. B. Amonette, D. S. D. Burke and R. D. Orr, Chem. Mater., 19, 5703 (2007).
B.-W. Zhu, T.-T. Lim and J. Feng, Chemosphere, 65, 1137 (2006).
L. Wu, M. Shamsuzzoha and S.M.C. Ritchie, J. Nanopart. Res., 7, 469 (2005).
United States Environmental Protection Agency, National water quality inventory, EPA 816-R-00-013; USEPA Office of Water: Washington, DC, August (2000).
H. H. Comly, J. Am. Med. Assoc., 129, 112 (1945).
P. J. Weyer, J. R. Cerhan, B. C. Kross, G. R. Hallberg, J. Kantamneni, G. Breuer, M. P. Jones, W. Zheng and C. Lynch, Epidemiology, 12, 327 (2001).
United States Environmental Protection Agency. National Primary Drinking Water Regulations: Contaminant Specific Fact Sheets, Inorganic Chemicals, Technical Version; 811-F-95-002a-T; USEPA Office of Water, Washington, DC, (1995). Korean Minister of Environment, Drinking Water Quality Management 5-3 (2011).
The EU Water Framework Directive, European Commission Environment (2000).
N. P. Cheremisinoff and P. N. Cheremisinoff, Carbon adsorption for pollution control, PTR Prentice Hall, 19 (1993).
G. N. Glavee, K. J. Klabunde, C.M. Sorensen and G. C. Hadjipanayis, Inorg. Chem., 34, 28 (1995).
L. S. Clesceri, A. E. Greenberg and A. D. Eaton, Standard methods for the examination of water and wastewater, 20th Ed., 4-108, American Public Health Association, Washington, DC (1998).
S. M. Ponder, J. R. Ford, J.G. Darab and T. E. Mallouk, Mater. Res. Soc. Sym. Proc., 556, 1269 (1999).
L. J. Matheson and P.G. Tratnyek, Environ. Sci. Technol., 28, 2045 (1994).
R.M. Cornell and U. Schwertmann, The iron oxides, VCH, Weinheim (1996).
R.G. Ford, P.M. Bertsch and J. C. Seaman, Clays Clay Miner., 45, 769 (1997).
J. P. Gaviría, A. Bohé, A. Pasquevich and D.M. Pasquevich, Physica B., 389, 198 (2007).
E. E. Carpenter, S. Calvin, R. M. Stroud and V. G. Harris, Chem. Mater., 15, 3245 (2003).
Y. Liu, S. A. Majetich, R. D. Tilton, D. S. Sholl and G.V. Lowry, Environ. Sci. Technol., 39, 1338 (2005).
J. T. Nurmi, P.G. Tratnyek, V. Sarathy, D. R. Baer, J. E. Amonette, K. Pecher, C. Wang, J.C. Linehan, D.W. Matson, J. C. Penn and M. D. Driessen, Environ. Sci. Technol., 39, 1221 (2005).
K. Sohn, S.W. Kang, S. Ahn, M. Woo and S. K. Yang, Environ. Sci. Technol., 40, 5514 (2006).
W. Yan, A. A. Herzing, C. J. Kiely and W. X. Zhang, J. Contam. Hydrol., 118, 96 (2010).
L. Signorini, L. Pasquini, L. Savini, R. Carboni, F. Boscherini, E. Bonetti, A. Giglia, M. Pedio, N. Mahne and S. Nannarone, Phys. Rev. B., 68, 195423-1–195423-8 (2003).
M. J. Alowitz and M.M. Scherer, Environ. Sci. Technol., 36, 299 (2002).
C. P. Huang, H.W. Wang and P. C. Chiu, Water Res., 32, 2257 (1998).
Y. H. H uang and T. C. Zhang, Water Res., 38, 2631 (2004).
R. Miehr, M.M. Tratnyek, J. Z. Bandstra, M. M. Scherer, M. J. Alowitz and E. J. Bylaska, Environ. Sci. Technol., 38, 139 (2004).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cho, M., Ahn, S. The influence of activated carbon support on nitrate reduction by Fe(0) nanoparticles. Korean J. Chem. Eng. 29, 1057–1062 (2012). https://doi.org/10.1007/s11814-011-0292-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-011-0292-1