Abstract
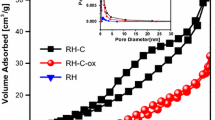
Microsilica, one kind of industrial solid waste material, was utilized firstly to prepare a carbon-microsilica composite adsorbent (CMS). The prepared adsorbent was characterized with XPS, SEM and Gas sorption experiments. The results indicated the SO3H groups, which are very effective in capturing cationic organic dye, were introduced onto the surface of CMS; the Brunauer-Emmett-Teller (BET) surface area (S BET ) and total pore volume (V total ) of CMS reach 51m2/g and 0.045 cm3/g, respectively. Meanwhile, the possibility of the utilization of the adsorbent for removal of methylene blue (MB) from aqueous solution was investigated. The effect of pH, contact time and initial MB concentration for MB removal were studied. Equilibrium data were modeled using the Langmuir, Freundlich and Dubinin-Radushkevich equations to describe the equilibrium isotherms. It was found that data fit to the Langmuir equation better than the Freundlich equation. Maximum monolayer adsorption capacity was calculated at different temperatures (298, 308, and 318 K) reach 251.81, 283.76 and 309.70 mg/g, respectively. It was observed that adsorption kinetics obeys the pseudo-first-order equation.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
M. S. Chiou, P.Y. Ho and H.Y. Li, Dyes Pigm., 60, 69 (2004).
A. S. Alzaydien, American J. Environ. Sci., 5, 197 (2009).
T. Robinson, G. McMullan, R. Marchant and P. Nigman, Bioresour. Technol., 77, 247 (2001).
G. Crini, Bioresour. Technol., 97, 1061 (2006).
A. A. Atia, A. M. Donia and W. A. Al-Amrani, Chem. Eng. J., 150, 55 (2009).
H. Hameed, A. L. Ahmad and K. N. A. Latiff, Dyes Pigm., 75, 143 (2007).
J. C. Park, J. B. Joo and J. Yi, Korean J. Chem. Eng., 26, 102 (2009).
G. V. Kumar, P. Ramalingam, M. J. Kim, C. K. Yoo and M. D. Kumar, Korean J. Chem. Eng., 27, 1025 (2010).
M. A. Rauf, S. M. Qadri, S. Ashraf and K. M. Al-Mansoori, Chem. Eng. J., 150, 90 (2009).
S. S. Azhar, A.G. Liew, D. Suhardy, K. F. Hafiz and M. D. Hatim, America J. Appl. Sci., 2, 1499 (2005).
F. Banat, S. Al-Asheh, S. Al-Anbar and S. Al-Refaie, Bull. Eng. Geol. Env., 66, 53 (2007).
Z.G. Hu, J. Zhang, W. L. Chan and Y. S. Szeto, Polymer, 47, 5838 (2006).
V. Vadivelan and K. V. Kumar, J. Colloid Interface Sci., 286, 90 (2005).
O. Hamdaoui, J. Hazard. Mater., 135, 264 (2006).
D. Chung, J. Mater. Sci., 37, 673 (2002).
D. Zhang, H. Luo, Y. Wang and H. Feng, Chem. Lett., 39, 424 (2010).
K. Kaneko, J. Membr. Sci., 96, 59 (1994).
X. Liang, M. Zeng and C. Qi, Carbon, 48, 1844 (2010).
M. Toda, A. Takagaki, M. Okamura, J. N. Kondo, S. Hayashi, K. Domen and M. Hara, Nature, 438, 178 (2005).
K. S.W. Sing, D. H. Everett, R. A.W. Haul, L. Moscou, R.A.W. Pierotti, J. Rouquérol and T. Siemieniewska, Pure Appl. Chem., 57, 603 (1985).
H. Z. Mousavi, A. Hosseynifar, V. Jahed and S. A. M. Dehghani, Braz. J. Chem. Eng., 27, 79 (2010).
D. Lu, Q. Cao, X. Cao and F. Luo, J. Hazard. Mater., 166, 239 (2009).
S. Lagergren, Handlingar, 24, 1 (1898).
Y. S. Ho and G. McKay, Chem. Eng. J., 70, 115 (1998).
I. Langmuir, J. Am. Chem. Soc., 40, 1361 (1918).
H. Freundlich, Phys. Chem., 57, 384 (1906).
P. A. M. Freitas, K. Iha, M. C. F. C. Felinto and M. E. V. Suárez-Iha, J. Colloid Interface Sci., 323, 1 (2008).
I. A. W. Tan, A. L. Ahmad and B. H. Hameed, J. Hazard. Mater., 164, 473 (2009).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, D., Ma, Y., Feng, H. et al. Removal of methylene blue from aqueous solution by a carbon-microsilica composite adsorbent. Korean J. Chem. Eng. 29, 775–780 (2012). https://doi.org/10.1007/s11814-011-0257-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-011-0257-4