Abstract
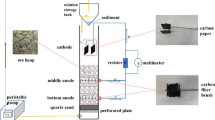
A process flowsheet was developed to recover copper metal from the lean sulfide ore of copper available at Malanjkhand, Hindustan Copper Limited (HCL), India. Copper pregnant leach solution (PLS) obtained from bio-heap leaching of chalcopyrite containing 0.3% copper was purified through solvent extraction (SX) and the copper recovered by electrowinning (EW). The copper-free raffinate obtained from SX stripping unit was returned back to the bioleaching circuit. The purity of the electrolytic copper produced at pilot scale was found to be 99.96%. During electrowinning, the effect of flow rate of electrolyte on current efficiency and energy consumption was also studied.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
M. Mishra, S. Singh, T. Das, R.N. Kar, K. S. Rao, L. B. Sukla and B. K. Mishra, Korean J. Chem. Eng., 25(3), 531 (2008).
S. C. Bouffard, Minerals Engineering, 15, 1115 (2008).
M. Dopson, L. Lovgren and D. Bostrom, Hydrometallurgy, 96, 288 (2009).
A. D. McMahon, Copper: A material Survey, Circular Bureau of Mines, U.S. Dept. of the Interior, Bureau of Mines, Washington (1964).
N. Pradhan, K. C. Nathsarma, K. S. Rao, L. B. Sukla and B. K. Mishra, Minerals Engineering, 21, 355 (2008).
R. L. Rich, Inorganic Reactions in Water 3e Ed. Springer, Berlin, 263 (2008).
A. Taylor, Copper heap leach design-Hit or Miss, ALTA Metallurgical Services (2002).
A. T. Bull, Korean J. Chem. Eng., 18, 137 (2001).
S. Mohapatra, C. Sengupta, B. D. Nayak, L. B. Sukla and B. K. Mishra, Korean J. Chem. Eng., 26(1), 108 (2009).
L.B. Sukla, K. C. Nathsarma, J. R. Mahanta, S. Singh, S. Behera, K. S. Rao, T. Subbaiah and B. K. Mishra, Korean J. Chem. Eng., 26, 1668 (2009).
L. Cifuentes, R. Ortiz and J.M. Casas, AIChE J., 51, 2273 (2005).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Panda, S., Sarangi, C.K., Pradhan, N. et al. Bio-hydrometallurgical processing of low grade chalcopyrite for the recovery of copper metal. Korean J. Chem. Eng. 29, 781–785 (2012). https://doi.org/10.1007/s11814-011-0254-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-011-0254-7