Abstract
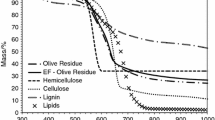
The kinetic analysis of pyrolysis process of longan seed was performed in a thermogravimetric analyzer. All experimental runs were carried out by using an initial sample mass of 15 mg and final temperature of 650 °C under the inert atmosphere of nitrogen. Particle sizes in the range from 0.05–2.1 mm and the heating rates from 5–100 °C/min were employed to investigate their effects on the thermogram and the kinetic parameters. The TG curves generally showed sigmoid shape and displayed one major peak in DTG curve. The main devolatilization of longan seed occurred over the temperature range of 210–330 °C. It was found that heat transfer resistance in a particle could be reduced either by decreasing the size of particle or increasing the heating rate. The thermal decomposition of longan seed could be well described by the two-parallel reactions kinetic model. This analysis of reaction kinetic gave the values of activation energy for the decomposition of the two fractions in the model corresponding closely to those of hemicellulose and lignin.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
S-S. Kim, F.A. Agblevor and J. Lim, J. Ind. Eng. Chem., 15, 247 (2009).
H. J. Park, Y.-K. Park, J.-I. Dong, J.-S. Kim, J.-K. Jeon, S.-S. Kim, J. Kim, B. Song, J. Park and K.-J. Lee, Fuel Process. Technol., 90, 186 (2009).
H. J. Park, J.-I. Dong, J.-K. Jeon, Y.-K. Park, K.-S. Yoo, S.-S. Kim, J. Kim and S. Kim, Chem. Eng. J., 143, 124 (2008).
Y.-H. Park, J. Kim, S.-S. Kim and Y.-K. Park, Bioresour. Technol., 100, 400 (2009).
H. J. Park, J.-I. Dong, J.-K. Jeon, K.-S. Yoo, J.-H. Yim, J. M. Sohn and Y.-K. Park, J. Ind. Eng. Chem., 13, 182 (2007).
H. I. Lee, H. J. Park, Y.-K. Park, J.Y. Hur, J.-K. Jeon and J.M. Kim, Catal. Today, 132, 68 (2008).
H. J. Park, J.-K. Jeon, J.M. Kim, H. I. Lee, J.-H. Yim, J. Park and Y.-K. Park, J. Nanosci. Nanotechnol., 8, 5439 (2008).
H. B. Goyal, D. Seal and R. C. Saxena, Renewable Sustainable Energy Rev., 12, 504 (2008).
S. Kim and Y. Eom, Korean J. Chem. Eng., 23, 409 (2006).
R. R. Baker, Thermochim. Acta, 23, 201 (1978).
J.A. Conesa, A. Marcilla, J. A. Caballero and R. Font, J. Anal. Appl. Pyrol., 58–59, 617 (2001).
J.A. Caballero, J.A. Conesa, R. Font and A. Marcilla, J. Anal. Appl. Pyrol., 42, 159 (1997).
H. Teng, H.-C. Lin and J.-A. Ho, Ind. Eng. Chem. Res., 36, 3947 (1997).
J. Guo and A. C. Lua, Biomass Bioenergy, 20, 223 (2001).
R. Font, A. Marcilla, E. Verdu and J. Devesa, J. Anal. Appl. Pyrol., 21, 249 (1991).
S. Junpirom, D.D. Do, C. Tangsathitkulchai and M. Tangsathitkulchai, Carbon, 43, 1936 (2005).
P. Luangkiattikhun, C. Tangsathitkulchai and M. Tangsathitkulchai, Bioresour. Technol., 99, 986 (2006).
K. Gergova, N. Petrov and S. Eser, Carbon, 32, 693 (1994).
T. Fisher, M. Hajaligol, B. Waymack and D. Kellogg, J. Anal. Appl. Pyrol., 62, 331 (2002).
H. Haykiri-Acma, J. Anal. Appl. Pyrol., 75, 211 (2006).
J. F. Gonzalez, J.M. Encinar, J. L. Canito, E. Sabio and M. Chacon, J. Anal. Appl. Pyrol., 67, 165 (2003).
A. J. Tsamba, W. Yang and W. Blasia, Fuel Process. Technol., 87, 523 (2006).
G. Varhegyi, M. J. Antal, E. Jakab and P. Szabo, J. Anal. Appl. Pyrol., 42, 73 (1997).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Junpirom, S., Tangsathitkulchai, C. & Tangsathitkulchai, M. Thermogravimetric analysis of longan seed biomass with a two-parallel reactions model. Korean J. Chem. Eng. 27, 791–801 (2010). https://doi.org/10.1007/s11814-010-0118-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-010-0118-6