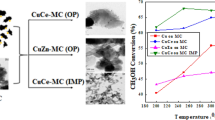
Abstract
The water gas shift (WGS) reaction has been investigated widely in fuel cell technologies due to the potential for high fuel efficiency and lower emissions during the production of pure hydrogen. Industrially, the WGS reaction occurs in one of the following two ways: (a) high-temperature in the range of 310–450°C with Fe-Cr catalyst, (b) low-temperature in the range of 210–250°C with Cu-ZnO-Al2O3. In this study, a mesoporous catalyst was prepared, with a large surface area and uniformity in both pore size and distribution, by using a one-pot synthesis method. The prepared CuO-CeO2-Al2O3 brought high CO conversion (82%), and was suitable for WGS reaction at low temperature (250 °C).
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
C. Zerva and C. J. Philippopoulos, Appl. Catal. B, 67, 105 (2006).
T. Tabakova, V. Idakiev, J. Papavasiliou, G. Avgouropoulos and T. Ioannides, Catal. Commun., 8, 101 (2007).
R. Burch, Phys. Chem. Chem. Phys., 8, 5483 (2006).
R. J. Gorte and S. Zhao, Catal. Today, 104, 18 (2005).
Y. Kim, P. Kim, C. Kim and J. Yi, J. Mater. Chem., 13, 2353 (2003).
Y. Kim, C. Kim, P. Kim and J. Yi, J. Non-Cryst. Solids, 351, 550 (2005).
I. Sobczak, J. Goscianska, M. Ziolek, J. Crams, C. Verrier, P. Bazin, O. Marie and M. Daturi, Catal. Today, 114, 281 (2006).
P. Kim, J. B. Joo, H. Kim, W. Kim, Y. Kim, I. K. Song and J. Yi, Catal. Lett., 104, 181 (2005).
D. R. Neuville, L. Cormier, V. Montouillout and D. Massiot, J. Non-Cryst. Solids, 353, 180 (2007).
M. T. Bore, H. N. Pham, T. L. Ward and A. K. Datye, Chem. Commun., 22, 2620 (2004).
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is dedicated to Professor Chang Kyun Choi for celebrating his retirement from the School of Chemical and Biological Engineering, Seoul National University.
Rights and permissions
About this article
Cite this article
Kim, JK., Kim, Y., Park, JW. et al. Preparation of CuO-CeO2-Al2O3 catalyst with mesopore structure for water gas shift reaction. Korean J. Chem. Eng. 26, 32–35 (2009). https://doi.org/10.1007/s11814-009-0006-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-009-0006-0