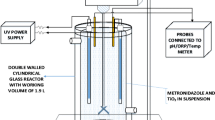
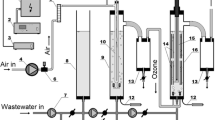
Abstract
The performance of both batch and continuous photo-catalytic reactors was studied to evaluate their capabilities in removing the sulfonyl urea herbicide of metsulfuron methyl (MM). It was found in a batch reactor that the addition of a small amount of powder activated carbon (PAC) significantly increased the rate of degradation of MM. The continuous photo-catalytic system resulted in 57% of MM removal. When a small dose of activated carbon was added in the photo-catalytic system, MM removal increased to 78–86% MM removal for retention times between of 5.25–21 min (corresponding to withdrawal rates of 10–40 mLmin−1). In this study, the pseudo first order rate constants of a continuous photo-catalytic system revealed that shorter retention times were associated with lower rate constants. Solid phase micro extraction/gas chromatography (SPME/GC) results showed that high concentrations of MM were broken down to small volatile organic compounds (VOCs) by photo-catalytic oxidation. PAC adsorbed the photo-products and increased the degradation of MM.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
K. M. Agbekodo, B. Legube and S. Dard, Water Research, 30(11), 2535 (1996).
R. Molinari, M. Borgese, E. Drioli, L. Palmisano and M. Schiavello, Catalysis Today, 75(1–4), 77 (2002).
W. Chu and C. C. Wong, Water Research, 38(4), 1037 (2004).
W. J. Masschelein and R. G. Rice, Ultraviolet light in water and wastewater sanitation, Lewis Publishers, Boca Raton, Florida (2002).
O. K. Scheible, M. C. Casey and A. Fondran, National technical information service, NITS Publication 86-145182 (1985).
M.V. Thampi and C. A. Sorber, Water Research, 21(7), 765 (1987).
E. Vulliet, C. Emmelin, J.-M. Chovelon, C. Guillard and J.-M. Herrmann, Environ. Chem. Lett., 1, 62 (2003)
A. Nathaporn, H. K. Shon, S. Vigneswaran and H. H. Ngo, Water Science and Technology: Water Supply, 6(2), 109 (2006).
V. Maurino, C. Minero, E. Pelizzetti and M. Vincenti, Colloids and Surfaces A: Physicochemical and Engineering Aspects, 151(1–2), 329 (1996).
G. C. Galletti, G. Chiavari, F. A. Mellon, and K. Parlsey, Journal of Analytical and Applied Pyrolysis, 21(1–2), 239 (1991).
E.M. Beyer, M. J. Duffy, J.V. Hay and D. D Schlueter, Chemistry, Degradation, and Mode of Action, 3, 117 (1987).
G. Mark, H.-G. Korth, H.-P. Schuchmann and C. von Sonntag, Journal of Photochemistry and Photobiology A: Chemistry, 101(2–3), 89 (1996).
C. Hu, T. Yuchao, L. Lanyu, H. Zhengping and T. Hongxiao, Journal of Chemical Technology and Biotechnology, 79, 247 (2004).
C. C. Wong and W. Chu, Chemosphere, 50(8), 981 (2003).
J. Arana, J. M. Dona-Rodriguez, E. Tello Rendon, C. Garriga i Cabo, O. Gonzalez-Diaz, J. A. Herrera-Melian, J. Perez-Pena, G. Colon and J. A. Navio, Applied Catalysis B: Environmental, 44(2), 161 (2003).
A. Bhattacharyya, S. Kawi and M. B. Ray, Catalysis Today, 98(3), 431 (2004).
A. Bianco Prevot, M. Vincenti, A. Bianciotto and E. Pramauro, Applied Catalysis B: Environmental, 22(2), 149 (1999).
Ö. E. Kartal, M. Erol and H. Ogïuz, Chemical Engineering & Technology, 24(6), 645 (2001).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Areerachakul, N., Vigneswaran, S., Ngo, H.H. et al. A continuous photocatalysis system in the degradation of herbicide. Korean J. Chem. Eng. 25, 663–669 (2008). https://doi.org/10.1007/s11814-008-0109-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-008-0109-z