Abstract
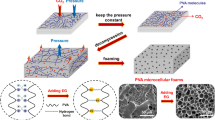
The production of microcellular plastic was studied in the polymethyl metacrylate (PMMA)-supercritical carbon dioxide and polycarbonate (PC)-supercritical carbon dioxide systems. The test pieces of PMMA and PC were put into a saturation vessel of which temperature and pressure were kept constant. Supercritical carbon dioxide at temperature between 303K and 393K and pressure between 100 bar and 250 bar was used as a foaming agent. After saturation of carbon dioxide, the pressure was quickly released to atmospheric pressure. The samples were immediately taken out from the vessel and heated in an oil bath. The fractured part of the sample was used for microstructure analysis with SEM. The effect of the saturation temperature, pressure of sorption and the foaming time on the cell mean size and cell density of the foam was investigated by considering the solubility of carbon dioxide in PMMA and PC. The foam morphologies of the foamed plastics were affected by solubility of carbon dioxide, which was directly related to saturation temperature and pressure. The cell density increased and, consequently, the cell size decreased with the solubility of carbon dioxide. The foaming time can be used a controlling factor to obtain the desired foam structure and the volume expansion ratio.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kwon, YK., Bae, HK. Production of microcellular foam plastics by supercritical carbon dioxide. Korean J. Chem. Eng. 24, 127–132 (2007). https://doi.org/10.1007/s11814-007-5022-3
Issue Date:
DOI: https://doi.org/10.1007/s11814-007-5022-3