Abstract
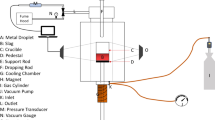
Wet limestone scrubbing is the most common flue gas desulfurization process (FGD) for control of sulfur dioxide emissions from the combustion of fossil fuels. Forced oxidation, which controls the overall reaction of the sulfur dioxide absorption, is the key path of the process. Manganese which comes from the coal is one of the catalysts during the forced oxidation process. In the present work, the two-film theory was used to analyze the sulfite forced oxidation reaction with an image boundary recognition technique, and the oxidation rate was experimentally studied by contacting pure oxygen with a sodium sulfite solution. There was a critical sulfite concentration 0.328 mol/L without catalyst or at a constant catalyst concentration value. The kinetics study focused on the active energy of the reaction and the reaction constant k; furthermore, we obtained the order with respect to the sulfite and Mn2+ concentrations. When the Mn2+ catalyst concentration was kept unchanged, the sulfite oxidation reaction rate was controlled by dual film and the reaction kinetics was first order with respect to sulfite while SO 2−3 concentration was below 0.328 mol/L; the sulfite oxidation reaction rate was controlled by gas film only and the reaction kinetics was zero order with respect to sulfite while SO 2−3 concentration over 0.328 mol/L. When SO 2−3 concentration was kept unchanged, the sulfite oxidation reaction rate depended on gas-liquid mass transfer and the reaction kinetics was different in various stages with respect to Mn2+ concentrations.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
V. Linek and V. Vacek, Chem. Engng. Sci., 36, 1747 (1981).
W. G. Whitman, Chem. and Met. Eng., 29, 147 (1923).
R. Higbie, Trans. Am. Inst. of Chem. Eng., 31, 365 (1935).
P. V. Danckwerts, Ind. Eng. Chem. Res., 43, 1460 (1951).
H. L. Toor and J. M. Marchello, AIChE J., 16, 513 (1970).
W. L. Weisnicht, L. Overman, C. C. Wang, H. L. Wang, J. Erwin and J. L. Hudson, Chem. Engng Sci., 35, 463 (1980).
W. Pasiuk-Bronikowska and T. Bronikowski, Chem. Engng Sci., 36, 215 (1981).
A. Jr. Huss, P. K. Lim and C. A. Eckert, J. Phys. Chem., 86, 4224 (1982).
W. Pasiuk-Bronikowska and J. Ziajka, Chem. Engng Sci., 40, 1567 (1985).
R. K. Ulrich, G. T. Rochelle and R. E. Prada, Chem. Engng Sci., 41, 2183 (1986).
T. Bronikowski and W. Pasiuk-Bronikowska, Chem. Engng Sci., 44, 1361 (1989).
J. Ziajka and W. Pasiuk-Bronikowska, Chem. Engng Sci., 44, 915 (1989).
A. Lancia, D. Musmarra, M. Prisciandaro and M. Tammaro, Chem. Engng. Sci., 54, 3019 (1999).
H. K. Lee, B. R. Deshwal and K. S. Yoo, Korean J. Chem. Eng., 22, 208 (2005).
B. Zhao, Y. Li, H. L. Tong, Y. Q. Zhuo, L. Zhang, J. Shi and C. H. Chen, Chem. Engng Sci., 60, 863 (2005).
H. L. J. Bäckström, J. Am. Chem. Soc., 49, 1460 (1927).
P. V. Danckwerts, Gas-liquid reaction, McGraw-Hill Publications, New York (1970).
G. C. Mishra and R. D. Srivastava, J. Appl. Chem. Biotechnol., 26, 401 (1976).
D. X. Shen, Z. Y. He and Y. R. Wang, Environ. Chem., 12, 99 (1993).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bo, Z., Yan, L., Yuqun, Z. et al. Mass transfer and kinetics study on the sulfite forced oxidation with manganese ion catalyst. Korean J. Chem. Eng. 24, 471–476 (2007). https://doi.org/10.1007/s11814-007-0082-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-007-0082-y