Abstract
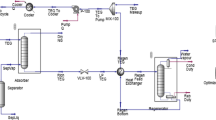
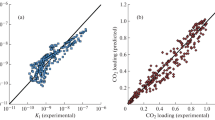
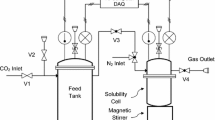
A new numerical model, which covers the full range of dehydration-plant operating conditions and wide range of experimental data results, estimates the amount of CH4, C2H6 and C3H8 absorbed per volume of triethylene glycol (TEG) circulated vs. the partial pressure of light alkanes and the absorber temperature. This article shows that the proposed numerical approach is more accurate than routine equation of states in predicting the solubility of light hydrocarbons in TEG. This article also provides comparisons between the results of the proposed model with experimental data and an equation of state results.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
S. W. Park, B. S. Choi and W. Lee, Korean J. Chem. Eng., 23, 138 (2006).
F.-Y. Jou, F. D. Otto and A. E. Mather, The Canadian J. Chem. Eng., 72, 130 (1994).
A. Bahadori and K. Zeidani, Oil & Gas Journal, 104, 27 (2006).
A. Bahadori, SPE No. 81121, Society of Petroleum Engineers, Latin American and Caribbean Petroleum Engineering Conference, Port-of-Spain, Trinidad, Apr. 27–30 (2003).
F.-Y. Jou, R. D. Deshmukh, F. D. Otto and A. E. Mather, Fluid Phase Equilibria, 36, 121 (1987).
P. L. Grizzle, SPE/EPA Exploration & Production Environmental Conference San Antonio, Texas, 7–10 March (1993).
C. O. Rueter, D. L. Reif, W. R. Menzies and R. J. M. Evans, SPE No. 29698 SPE Advanced Technology Series, 4 (1993).
S. A. Ahmad and A. Khanna, Korean J. Chem. Eng., 20, 736 (2003).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bahadori, A. New numerical model for solubility of light alkanes in triethylene glycol. Korean J. Chem. Eng. 24, 418–425 (2007). https://doi.org/10.1007/s11814-007-0072-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-007-0072-0