Abstract
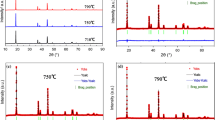
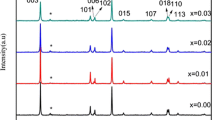
A lithium insertion material having the composition LiNi0.3Co0.3Mn0.3Fe0.1O2 was synthesized by simple sol-gel method. The structural and electrochemical properties of the sample were investigated using X-ray diffraction spectroscopy (XRD) and the galvanostatic charge-discharge method. Rietvelt analysis of the XRD patterns shows that this compound can be classified as α-NaFeO2 structure type (R3m; a=2.8689(5) Å and 14.296(5) Å in hexagonal setting). Rietvelt fitting shows that a relatively large amount of Fe and Ni ion occupy the Li layer (3a site) and a relatively large amount of Li occupies the transition metal layer (3b site). LiNi0.3Co0.3Mn0.3Fe0.1O2 when cycled in the voltage range 4.3–2.8 V gives an initial discharge capacity of 120 mAh/g, and stable cycling performance. LiNi0.3Co0.3Mn0.3Fe0.1O2 in the voltage range 2.8–4.5 V has a discharge capacity of 140 mAh/g, and exhibits a significant loss in capacity during cycling. Ex-situ XRD measurements were performed to study the structure changes of the samples after cycling between 2.8–4.3 V and 2.8–4.5 V for 20 cycles. The XRD and electrochemical results suggested that cation mixing in this layered structure oxide could be causing degradation of the cell capacity.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
S.-H. Kang, J. Kim, M. E. Stroll, D. Abraham, Y. K. Sun and K. Amine, J. Power Sources, 112, 41 (2002).
H. Arai, S. Okada, Y. Sakurai and J. Yamaki, Solid State Ionics, 95, 275 (1997).
T. Ohzuku, A. Ueda and M. Nagayams, J. Electrochem. Soc., 140, 1862 (1993).
T. Ohzuku, A. Ueda and M. Kouguchi, J. Electrochem. Soc., 142, 4033 (1995).
G. Vitins and K. West, J. Electrochem. Soc., 144, 2587 (1997).
Y.-I. Jang, B. Huang, Y.-M. Chaiang and D.R. Sadoway, Electrochem. Solid-State Lett., 1, 13 (1998).
B. Ammundsen and J. Paulsen, Adv. Mater., 13, 943 (2001).
R. J. Gummow and M. M. Thackeray, J. Electrochem. Soc., 141, 1178 (1994).
Y. Makimura and T. Ohzuku, J. Power Sources, 119–121, 156 (2003).
N. Yabuuchi and T. Ohzuku, J. Power Sources, 119–121, 171 (2003).
T. Ohzuku and Y. Makimura, Chem. Lett., 642 (2001).
N. Yabuuchi and T. Ohzuku, J. Power Sources, 119–121, 171 (2003).
Y. S. Meng, Y. W. Wu, B. J. Hwang, Y. Li and G. Ceder, J. Electrochem. Soc., 151, A1134 (2004).
C. Wolverton and A. Zunger, J. Electrochem. Soc., 145, 2424 (1998).
A. Rougier, P. Gravereau and C. Delmas, J. Electrochem. Soc., 143, 1168 (1996).
J. Baker, R. Koksbang and M. Y. Saidi, Solid State Ionics, 89, 25 (1996).
J.-M. Kim and H.-T. Chung, Electrochem. Acta, 49, 937 (2004).
G. T. K. Fey, J. G. Chen, V. Subramanian and T. Osaka, J. Power Sources, 384, 112 (2002).
T. Ohzuku and Y. Makihara, Chem. Lett., 642 (2001).
S. H. Na, H. S. Kim and S. I. Moon, Solid State. Ionics., 176, 313 (2005).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Son, JT., Cairns, E. Structure and electrochemical characterization of LiNi0.3Co0.3Mn0.3Fe0.1O2 cathode for lithium secondary battery. Korean J. Chem. Eng. 24, 888–891 (2007). https://doi.org/10.1007/s11814-007-0060-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-007-0060-4