Abstract
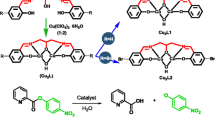
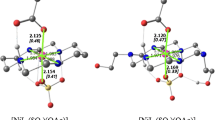
Two micellized Cu(II) and Fe(III) ion complexes of octadecyl diethylene tri-1-amide are used as nucleophilic reagents in the cleavage of the phosphate ester paraoxon. These complexes form metallomicelles, which coordinate with the paraoxon phosphorous (chemically similar to the nerve agent sarin). Association colloids (e.g., micelles, microemulsions, or vesicles) increase the rate of the nucleophilic reaction. Hence, it is reasonable to examine the hydrolysis process of paraoxon in aqueous metallomicelles. As micelles form and incorporate reactants, the rate constant should change; therefore, the rate constant should be affected by the critical micelle concentrations (CMC) of Cu(II) and Fe(III) complexes. The concentrations tested were the CMC value, two values lower, and two values higher. The CMC was found to be the most reactive concentration. The kinetic data (half-life times) for paraoxon degradation were 6.1 and 28.8 min in the presence of the Cu(II) and the Fe(III) metallomicelle complex, respectively. The stability constants of the metal complexes showed large values that denoted significant stability for both complexes.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Abbreviations
- CMC:
-
critical micelle concentration
- FTIR:
-
Fourier transform infrared
References
Menger, F.M., L.H. Gan, E. Johnson, and D.H. Durst, Phosphate Ester Hydrolysis Catalyzed by Metallomicelles, J. Am. Chem. Soc. 109:2800 (1987).
Scrimin, P., G. Ghirlanda, P. Tecilla, and R.A. Moss, Comparative Reactivities of Phosphate Ester Cleavages by Metallomicelles, Langmuir 12:6235 (1996).
Mancin, F., P. Tecilla, and U. Tonellato, Metallomicelles Made of Ni(II) and Zn(II) Complexes of 2-Pyridinealdoxime-Based Ligands as Catalyst of the Cleavage of Carboxylic Acid Esters, Langmuir 16:227 (2000).
Zheng, F., C.-G. Zhan, and R.L. Ornstein, Theoretical Studies of Reaction Pathways and Energy Barriers for alkaline Hydrolysis of Phosphotriesterase Substrates Paraoxon and Related Toxic Phosphofluoridate Nerve Agents, J. Chem. Soc. Perkin. Trans. 2:2355 (2001).
Shaw, R.W., and M.J. Cullinane, Destruction of Military Toxic Materials, in Encyclopedia of Environmental Analysis and Remediation, edited by R.A. Myers, John Wiley & Sons, New York, 1998, Vol. 5, p. 2821.
Moss, R.A., and Y. Ihara, Cleavage of Phosphate Esters by Hydroxyl-Functionalized Micellar and Vesicular Reagents, J. Org. Chem. 48:588 (1983).
Siqing, C., Z. Xiancheng, M. Xiangguang, and Y. Xiaoqi, Metallomicellar Catalysis Hydrolysis of p-Nitrophenyl Picolinate Catalyzed by Copper(II), Nickel(II), and Zinc(II) Complexes of Long Alkyl Pyridine Ligands in Micellar Solution, J. Colloid Interface Sci. 224:333 (2000).
Koča, J., C.-G. Zhan, R.C. Rittenhouse, and R.L. Ornstein, Mobility of the Active Site Bound Paraoxon and Sarin in Zinc-Phosphotriesterase by Molecular Dynamics Simulation and Quantum Chemical Calculation, J. Am. Chem. Soc. 123:817 (2001).
Kimura, E., H. Hashimoto, and T. Koike, Hydrolysis of Lipophilic Esters Catalyzed by a Zinc(II) Complex of a Long Alkyl-Pendant Macrocyclic Tetraamine in Micellar Solution, J. Am. Chem. Soc. 118:10963 (1996).
Yang, Y.-C., Chemical Detoxification of Nerve Agent VX, Acc. Chem. Res. 32:109 (1999).
Bunton, C.A., Chemical Warfare, in Macmillan Encyclopedia of Chemistry, edited by J.J. Lagowski, Simon & Schuster/Macmillan, New York, 1997, Vol. 1, p. 343.
Defrank, J.J., Organophosphorous Cholinesterase Inhibitors: Detoxification by Microbial Enzymes, in Applications of Enzyme Biotechnology, edited by J.W. Kelly and T.O. Baldwin, Plenum Press, New York, 1991, p. 165.
Heilbronn-Wikstrom, E., Phosphorylated Cholinesterase: Their Formation Reactions and Hydrolysis, Sven. Kem. Tidskr. 77:598 (1965).
Moss, R.A., A.T. Kotchevar, B.D. Park, and B.D. Scrimin, Comparative Reactivities of Phosphotriesters Toward Iodosocarboxylates in Cationic Micelles, Langmuir 12:2200 (1996).
Lion, C., J.P. Boukou-Poba, and M. Hedayatullah, Nouveaux décontaminants. Action du Monoperphtalate de Magnesium (MPPM) sur Quelques insecticides et toxiques de Guerre Organo Phosphorees ou Organosulfers, Phosphorus Sulfur Silicon Relat. Elem. 56:213 (1991).
Lion, C., M. Hedayatullah, P. Bauer, J.P. Boukou-Poba, and C. Charvy, Nouveaux décontaminants. Destruction de Toxiques organophosphorés ou Soufres par des Peracides Mono-, Bi- et Tricycliques saturés, Bull. Soc. Chim. Belg. 101:249 (1992).
Moss, R.A., and H. Morales-Rojas, Kinetics of Cleavage of Thiophosphates and Phosphonothioates by Miceller Iodosocarboxylates and Copper Metallomicelles, Langmuir 16:6485 (2000).
Bowers, J., M.J. Danks, D.W. Bruce, and R.K. Heenan, Surface and Aggregation Behavior of Aqueous Solutions of Ru(II) Metallosurfactants: 1. Micellization of [Ru(bipy)2(bipy′)][Cl]2 Complexes, Langmuir 19:292 (2003).
Bowers, J., M.J. Danks, D.W. Bruce, and J.R.P. Webster, Surface and Aggregation Behavior of Aqueous Solutions of Ru(II) Metallosurfactants: 2. Adsorbed Films of [Ru(bipy)2(bipy′)][Cl]2 Complexes, Langmuir 19:299 (2003).
Johnson, J.Y., Improvement in the Manufacture and Production of Wetting, Cleansing, Dispersing and Like Agents, British Patent 378,383 (1932).
Badawi, A.M., A.A. Hafiz, and H.A. Ibrahim, Catalytic Destruction of Malathion by Metallomicelle Layers, J. Surfact. Deterg. 6:239 (2003).
Hafiz, A.A., M.Y. El Awadi, A.M. Badawi, and S.M. Mokhtar, Catalytic Destruction of Paraoxon by Metallomicelle Layers of Co(II) and Cr(III), J. Surfact. Deterg. 8:203 (2005).
Sawyer, D., W.E. Heineman, and J. Beebe, Chemistry Experiments for Instrumental Methods, John Wiley & Sons, New York, 1984, p. 4241.
Lee, J.D., Concise Inorganic Chemistry, 3rd edn., Van Nostrand Reinhold, London, 1983, p. 103.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Hafiz, A.A. Metallosurfactants of Cu(II) and Fe(III) complexes as catalysts for the destruction of paraoxon. J Surfact Deterg 8, 359–363 (2005). https://doi.org/10.1007/s11743-005-0369-8
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11743-005-0369-8