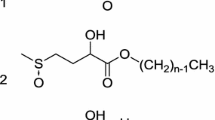
Abstract
Several novel and some previously known, mostly sugar-based, surfactants have been synthesized and some of their surface properties have been characterized and compared with those of commercial nonylphenol ethoxylates. The surfactant solubility in water, ethanol, and dodecane was studied. The properties of these compounds as emulsification agents in systems composed of the surfactant with water/isopropyl myristate, water/rapeseed oil, and water/dodecane are presented. The aqueous solubility of the surfactants follows the general trend expected from their hydrophilic-lipophilic balance according to Griffin (HLBG), but it is also clear that the nature of the headgroup and the structure of the nonpolar part affect the solubility in a manner not aptured in the standard HLBG concept. An ester or amine group as the connecting unit between the hydrophile and the hydrophobe produces a more water-soluble surfactant than the corresponding amide derivative. Some effective emulsifiers were found. For instance, the surfactants with a dehydroabietic nonpolar group appear to be promosing emulsifiers. Most sugar-based surfactants were able to form macroemulsions of up to around 2 wt/vol% of oil. The stability of many of these amulsions was very high, extending for months.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Abbreviations
- 2D:
-
two-dimensional
- DMSO:
-
dimethylsulfoxide
- HLB:
-
hydrophilic-lipophilic balance
- HLBd:
-
HLB according to Davies
- HLBG:
-
HLB according to Griffin
- IR:
-
infrared
- lit.:
-
literature
- MeOH:
-
methanol
- m.p.:
-
melting point
- MW:
-
molecular weight
- NMR:
-
nuclear magnetic resonance
- NP-6, NP-10, and NP-20:
-
nonylphonolethoxylates of 6, 10, and 20 ethylene oxide units
- O/W:
-
oil in water
- PEG:
-
polyethyelene glycol
- PTT:
-
phase inversion temperature
- W/O:
-
water in oil
References
von Rybinsky, W., Alkyl Glycosides and Polyglycosides, Current Opin. Colloid Interface Sci. 1:587 (1996).
Karsa, D.R., and M.R. Porter (eds.), Biodegradability of Surfactants, Blackie & Sons, London, 1995.
The Manufacturing Confectione, in McCutcheon’s Emulsifiers and Detergents, International Edn., Glen Rock, NJ, 1986.
Huford, J.R., in Developments in Food Carbohydrate 2, edited by C.K. Lee, Applied Science Publishers, London, 1980, p. 327.
Andree, H., and B. Middelhauve, Möglichkeiten des Einsatzes von Alkylpolyglucosiden in Wasch- und Spülmittein, Tenside Surfactants Deterg. 28:413 (1991).
Balzer, D., Alkylpolyglucosides, Their Physico-chemical Properties and Their Uses, Tenside Surfactants Deterg. 28:419 (1991).
Internal Report, Akzo Nobel Surface Chemistry, Stenungsund, Sweden, 1997.
Pes, M.A., K. Aramaki, N. Nakamura, and K. Hironobu, Temperature-Insensitive Microemulsions in a Sucrose Monoalkanoate System. J. Colloid Interface Sci. 178:666 (1996).
Jönsson, B., B. Lindman, K. Holmberg, and B. Kronberg, Introduction to Surfactants, in Surfactant and Polymers in Aqueous Solution, 2nd edn., John Wiley & Sons, Chichester, 2002, p. 24.
Kameyama, K., and T. Takagi, Micellar Properties of Octylglucoside in Aqueous Solutions, J. Colloid Interface Sci. 137:1 (1990).
Waltermo, Å., P.M. Claesson, E. Manev, S. Simonsson, I. Johansson, and V. Bergeron, Foam and Thin-Liquid-Film Studies of Alkyl Glucoside Systems, Langmuir 12:5271 (1996).
Matsumura, S., K. Imai, and S. Yoshikawa, Surface Activities, Biodegradability and Antimicrobial Properties of n-Alkyl Glucosides, Mannosides, and Galactosides, J. Am. Oil Chem. Soc. 67:996–1001 (1990).
Piispanen, P.S., An Improved Method of Synthesis of 2-Alkylamino-2-deoxy-d-glucopyranose and 1,2-Dialkylamino-1,2-dideoxy-d-(N)-β-glucoside. J. Org. Chem. 68:628 (2003).
Piispanen, P.S., Synthesis and Characterization of Surfactants Based on Natural Products, Ph.D. Thesis, KTH, Stockholm, Sweden, 2002, Thesis is also available at: http://media.lib.kth.se:8080/dissengrefhit.asp?dissnr=3383.
Mehltretter, C.L., and J.C. Rankin, Gluconamides, U.S. Patent 2,662,073 (1953).
Matero, A, Å. Mattson, and M. Svensson, Alkyl Polyglucosides as Hydrotropes, J. Surfact. Deterg. 4:1 (1998).
Piispanen, P.S., U.R.M. Kjellin, B. Hedman, and T. Norin, Synthesis and Surface Measurements of Surfactants Derived from Dehydroabietic Acid, Ibid. 6:125–130 (2002).
Wafina, M.G., and N.V. Molodtsov, Synthesis of Some p-Nitrophenyl 2-Acylamino-2-deoxy-d-glucosides and Their Hydrolysis with the β-d-Hexosaminidase from Hohenbuehelia serotina, Carbohydr. Res. 47:188 (1976).
Zhang, T., and R.E. Marchant, Novel Polysaccharide Surfactants: The Effect of Hydrophobic and Hydrophilic Chain Length on Surface-Active Properties. J. Colloid Interface Sci. 177:419 (1996).
Piispanen, P.S., B. Hedman, and T. Norin, Synthesis and Characterization of Dehydroabietic Acid Derivatives Suitable for Surfactant Synthesis, J. Surfact. Deterg. 5:165 (2002).
Fernandez-Bolaños, J., J. Galbis Perez, and F. Zamora Mata, An. Quim. 79:206 (1983).
Heyns, K., R. Eichstedt, and K.-H. Meinecke, Die Umsetzung von Fructose and Sorbose mit Ammoniak und Aminen, Chem. Ber. 88:1551 (1955).
Griffin, W.C., Calculation of HLB values of Non-ionic Surfactants. J. Soc. Cosmet. Chem. 5:249 (1954).
Davies, J.T. Interfacial Phenomena, Academic Press, New York, 1961.
Penzel, E., and K. Oppenländer, Charakterisierung einiger nichtionoger Emulgatoren: Einige kritische Anmerkungen zum HLB-system, Tenside Deterg. 3:129 (1974).
Davies, J.T.: A Quantitative Theory of Emulsion Type, I. Physical Chemistry of the Emulsifying Agent, Proc. 2nd Int. Congr. Surface Act. 1:426 (1957).
Waltermo, Å., M. Sjöberg, B. Anhede, and P.M. Claesson, Adsorption of an Ethoxylated Amine Surfactant on Mica and Its Effect on the Surface Forces, J. Colloid Interface Sci. 156:365 (1993).
Bancroft, W.D., The Theory of Emulsification V, J. Phys. Chem. 17:501 (1913).
Bancroft, W.D., The Theory of Emulsification VI, J. Phys. Chem. 19:275 (1915).
Syper, L., K.A. Wilk, A. Sokolowski, and B. Burczyk, Synthesis and Surface Properties of N-Alkylaldonamides, Progr. Colloid Polym. Sci. 119:81 (1998).
Lide, D.R., Handbook of Chemistry and Physics 1992–93, CRC Press, Boca Raton, FL, 1992, p. 42.
Shinoda, K., A. Carlsson, and B. Lindman, On the Importance of Hydroxyl Groups in the Polar Headgroup of Nonionic Surfactants and Membrane Lipids, Adv. Colloid Interface Sci. 64:253 (1996).
Kabalnov, A., and H. Wennerström, Macroemulsion Stability: The Oriented Wedge Theory Revisited, Langmuir 12:276 (1996).
Shinoda, K., and H. Saito, The Effect of Temperature on the Phase Equilibria and the Types of Dispersions of the Ternary System Composed of Water. Cyclohexane, and Nonionic Surfactant, J. Colloid Interface Sci. 26:70 (1968).
Shinoda, K., and H. Saito, The Stability of O/W Emulsions as Functions of Temperature and the HLB of Emulsifiers: The Emulsification by PIT-Method, Ibid. 30:258 (1969).
Shinoda, K., and H. Sagitani, Emulsifier Selection in Water/Oil Type Emulsions by the Hydrophile-Lipophile Balance-Temperature System, Ibid. 64:68 (1978).
Saito, H., and K. Shinoda, The Stability of W/O Type Emulsions as a Function of Temperature and of the Hydrophilic Chain Length of the Emulsifier, Ibid. 32:647 (1970).
Davis, H.T., Factors Determining Emulsion Type: Hydrophile-Lipophile Balance and Beyond, Colloids Surf. A. 91:9 (1994).
Shinoda, K., and S. Friberg, Emulsions and Solubilization, John Wiley & Sons, New York, 1986, 174 pp.
Griffin, W.C., Classification of Surface-Active Agents by HLB, J. Soc. Cosmet. Chem. 1:311 (1949).
Jönsson, B., B. Lindman, K. Holmberg, and B. Kronberg, Surfactants and Polymers in Aqueous Solution, reprint, John Wiley & Sons, Chichester, England, 2002, p. 355.
Salager, J.L., Formulation Concepts for the Emulsion Maker, in Pharmaceutical Emulsions and Suspensions, edited by F. Nielloud and G. Marti-Mestres, Marcel Dekker, New York, 1989, p. 20.
Kahl, H., K. Quitzsch, and E.H. Stenby, Phase Equilibria of Microemulsion Forming System n-Decyl-β-d-glucopyranoside/Water/n-Octane/1-Butanol, Fluid Phase Equilibria 13:295 (1997).
Jacobs, B., T. Sottmann, and R. Strey, Efficiency Boosting with Amphiphilic Block Copolymers—A New Approach to Microemulsion Formulation, Tenside Surfactants Deterg. 37:356 (2000).
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Piispanen, P.S., Persson, M., Claesson, P. et al. Surface properties of surfactants derived from natural products. Part 1: Syntheses and structure/property relationships—Solubility and emulsification. J Surfact Deterg 7, 147–159 (2004). https://doi.org/10.1007/s11743-004-0298-6
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11743-004-0298-6