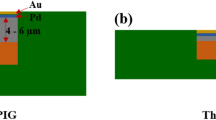Abstract
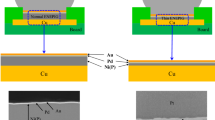
Using an electroless nickel/electroless palladium/immersion gold (ENEPIG) surface finish with a thick palladium–phosphorus (Pd-P) layer of 1 μm, the intermetallic compound (IMC) growth between the ENEPIG surface finish and lead-free solders Sn-3.5Ag (SA) or Sn-3.0Ag-0.5Cu (SAC) after reflow soldering and during solid-state aging at 150°C was investigated. After reflow soldering, in the SA/ENEPIG and SAC/ENEPIG interfaces, thick PdSn4 layers of about 2 μm to 3 μm formed on the residual Pd-P layers (~ 0.5 μm thick). On the SA/ENEPIG interface, Sn was detected on the upper side of the residual Pd-P layer. On the SAC/ENEPIG interface, no Sn was detected in the residual Pd-P layer, and Cu was detected in the interface between the Pd-P and PdSn4 layers. After 300 h of aging at 150°C, the residual Pd-P layers had diffused completely into the solders. In the SA/ENEPIG interface, an IMC layer consisting of Ni3Sn4 and Ni3SnP formed between the PdSn4 layer and the nickel–phosphorus (Ni-P) layer, and a (Pd,Ni)Sn4 layer formed on the lower side of the PdSn4 layer. On the SAC/ENEPIG interface, a much thinner (Pd,Ni)Sn4 layer was observed, and a (Cu,Ni)6Sn5 layer was observed between the PdSn4 and Ni-P layers. These results indicate that Ni diffusion from the Ni-P layer to the PdSn4 layer produced a thick (Pd,Ni)Sn4 layer in the SA solder case, but was prevented by formation of (Cu,Ni)6Sn5 in the SAC solder case. This causes the difference in solder joint reliability between SA/ENEPIG and SAC/ENEPIG interfaces in common, thin Pd-P layer cases.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Y. Oda, M. Kiso, S. Kurosaka, A. Okada, K. Kitajima, and S. Hashimoto, in Proceeding of International Microelectronics and Packaging Society, IMAPS (2008).
W.H. Wu, C.S. Lin, S.H. Huang, and C.E. Ho, J. Electron. Mater. 38, 2387 (2010).
S. Nakahara and R.J. McCoy, Thin Solid Films 88, 285 (1982).
G. Sharma, C.M. Eichfeld, and S.E. Mohney, J. Electron. Mater. 32, 1209 (2003).
Y.M. Kim, J.Y. Park, and Y.H. Kim, J. Electron. Mater. 41, 763 (2012).
K. Masui and M. Kajihara, J. Alloys Compd. 485, 144 (2009).
V. Vuorinen, T. Laurila, H. Yu, and J.K. Kivilahti, J. Appl. Phys. 99, 3530 (2006).
C.E. Ho, L.H. Hsu, S.W. Lin, and M.A. Rahman, J. Electron. Mater. 41, 2 (2012).
C.P. Lin and C.M. Chen, J. Alloys Compd. 547, 37 (2013).
C.E. Ho, T.T. Kuo, C.C. Wang, and W.H. Wu, Electron. Mater. Lett. 8, 495 (2012).
C.E. Ho, W.Z. Hsieh, C.H. Yang, T.C. Yeh, and T.T. Kuo, J. Electron. Mater. 44, 568 (2015).
D.G. Kim and S.B. Jung, Mater. Trans. 46, 1295 (2005).
C.C. Chi and T.H. Chuang, J. Electron. Mater. 35, 471 (2006).
C.W. Hwang, M. Kiso, S. Hashimoto, and K. Suganuma, J. Mater. Res. 18, 2540 (2003).
M.A. Rahman, C.E. Ho, W. Gierlotka, and J.C. Kuo, J. Electron. Mater. 43, 4582 (2014).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Oda, Y., Fukumuro, N. & Yae, S. Intermetallic Compound Growth between Electroless Nickel/Electroless Palladium/Immersion Gold Surface Finish and Sn-3.5Ag or Sn-3.0Ag-0.5Cu Solder. J. Electron. Mater. 47, 2507–2511 (2018). https://doi.org/10.1007/s11664-018-6067-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11664-018-6067-2