Summary
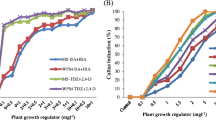
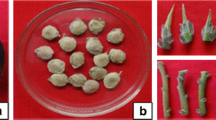
Biotechnology has offered a nonconventional method of plant propagation and has been intensively applied as a conservation strategy for sustaining biodiversity for rare plants. In vitro conservation through micropropagation of Ochreinauclea missionis, a rare, endemic and medicinal tree species of Western Ghats in Karnataka region of India is reported. Multiple shoots were initiated from nodal explants on Murashige and Skoog (MS) medium supplemented with 8.8 μM 6-benzylaminopurine (BA) and 0.3% (w/v) activated charcoal. Shoots were elongated in MS medium with a combination of 2.2 μM BA and 5.3 μM α-naphthaleneacetic acid (NAA) or growth regulator-free medium. Individual shoots with a minimum of one node were excised and rooted in vitro on MS medium with 0.3% activated charcoal or ex vitro rooted by treatment with 49 μM indole-3-butyric acid (IBA) for 30 min. Regenerants acclimated in Soil-rite exhibited 65% survival in the greenhouse.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Anonymous World plant conservation bibliography, London: Kew Royal Botanic Gardens/World Conservation Monitoring Centre; 1990:645 pp.
Augustine, A. C.; D'Souza, L. Micropropagation of an endangered forest tree Zanthoxylum rhetsa Roxb. Phytomorphology 47:319–323; 1997.
Fridborg, G.; Pedersen, M.; Landstrom, C. E.; Eriksson, J. The effect of activated charcoal on tissue cultures, adsorption of metabolites inhibiting morphogenesis. Physiol. Plant. 43:104–106; 1978.
George, E. F.; Sherrington, P. D. Plant propagation by tissue culture: handbook and directory of commercial laboratories. London: Exegetics Ltd.; 1984:102–110.
Kirtikar, K. R.; Basu, B. D. Indian medicinal plants. Delhi: Jayyed Press; 1975:1249–1250.
Lakshmanan, P.; Lee, C. L.; Goh, C. J. An efficient in vitro method for mass propagation of a woody ornamental Ixora coccinea L. Plant Cell Rep. 16:572–577; 1997.
Liew, T. K.; Chan, L. K.; Chris, T. K. H. In vitro rooting of Sentang shoots (Azadirachta excelsa) and acclimatisation of the plantlets. In Vitro Cell Dev. Biol. Plant 35:396–401; 1999.
Lynch, P. T. Tissue culture techniques in in vitro plant conservation. In: Benson, E. E., ed. Plant conservation biotechnology. London: Taylor & Francis Ltd.; 1999:41–62.
Moura-Costa, P. H.; Viana, A. M.; Mantell, S. H. In vitro plantlet regeneration of Ocotea catharinensis, an endangered Brazilian hardwood forest tree. Plant Cell Tiss. Organ Cult. 35:279–286; 1993.
Murashige, T.; Skoog, F. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant. 15:473–497; 1962.
Nayar, M. P.; Sastry, A. R. K. Red data books of Indian Plants, vol. 3. Calcutta: Botanical Survey of India Publications; 1990:222–223.
Preece, J. E.; Compton, M. E. Problems with explant exudation in micropropagation. In: Bajaj, Y. P. S., ed. Biotechnology in agriculture and forestry, vol. 17, High-tech and micropropagation I. Berlin, Heidelberg: Springer-Verlag; 1991:168–186.
Siril, E. A.; Dhar, U. Micropropagation of mature Chinese tallow tree (Sapium schiferum Roxb). Plant Cell Rep. 16:637–640; 1997.
Thorpe, T. A.; Biondi, S. Conifers. In: Sharp, W. R.; Evans, D. A.; Ammirato, P. V.; Yamada, T., eds. Handbook of plant cell cultures, vol. 2, New York: Macmillan; 1984:435–470.
Tisserat, B. Embryogenesis, organogenesis and plant regeneration. In: Dixon, R. A., ed., Plant cell culture, a practical approach, Oxford: IRC; 1985:79–105.
Weatherhead, M. A.; Burdon, L.; Henshaw, G. G. Some effects of activated charcoal as an additive to plant tissue culture media. Z. Pflanzenphysiol. 89:141–147; 1978.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dalal, N.V., Rai, R.V. in vitro propagation of Ochreinauclea missionis (Wall. EX G. Don), an ethnomedicinal endemic and threatened tree. In Vitro Cell.Dev.Biol.-Plant 37, 820–823 (2001). https://doi.org/10.1007/s11627-001-0136-5
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11627-001-0136-5