Abstract
Objective
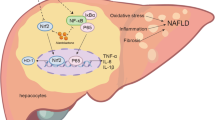
Berberine (BBR) has emerged as a promising therapeutic agent for nonalcoholic fatty liver disease (NAFLD). This study aims to elucidate the underlying molecular mechanisms.
Methods
In this study, db/db mice were chosen as an animal model for NAFLD. A total of 10 healthy C57BL/6J mice and 30 db/db mice were randomly allocated to one of 4 groups: the normal control (NC) group, the diabetic control (DC) group, the Metformin (MET) therapy group, and the BBR therapy group. The total cholesterol (TC), triacylglycerol (TG), low-density lipoprotein cholesterol (LDL-c), high-density lipoprotein cholesterol (HDL-c), aspartate aminotransferase (AST) and alanine aminotransferase (ALT) levels in the serum were measured. The glutathione peroxidase (GSH-Px), glutathione (GSH), malondialdehyde (MDA), superoxide dismutase (SOD), catalase (CAT), interleukin (IL)-1β, tumor necrosis factor (TNF)-α and monocyte chemotactic protein 1 (MCP-1) levels in liver tissue were measured. Hematoxylin and eosin (H&E), acid-Schiff (PAS) and TUNEL stanning was performed for histopathological analysis. Western blotting and immunohistochemistry were conducted to detect the expression levels of key proteins in the AMPK/SIRT1 pathway.
Results
BBR could improve lipid metabolism, attenuate hepatic steatosis and alleviate liver injury significantly. The excessive oxidative stress, high levels of inflammation and abnormal apoptosis in db/db mice were reversed after BBR intervention. BBR clearly changed the expression of AMP-activated protein kinase (AMPK)/Sirtuin 1 (SIRT1), and their downstream proteins.
Conclusion
BBR could reverse NAFLD-related liver injury, likely by activating the AMPK/SIRT1 signaling pathway to inhibit oxidative stress, inflammation and apoptosis in hepatic tissue.
Article PDF
Similar content being viewed by others

Avoid common mistakes on your manuscript.
References
Younossi ZM. Non-alcoholic fatty liver disease–A global public health perspective. J Hepatol, 2019,70(3):531–544
Riazi K, Azhari H, Charette JH, et al. The prevalence and incidence of NAFLD worldwide: a systematic review and meta-analysis. Lancet Gastroenterol Hepatol, 2022,7(9):851–861
Pastori D, Sciacqua A, Marcucci R, et al. Prevalence and Impact of Nonalcoholic Fatty Liver Disease in Atrial Fibrillation. Mayo Clin Proceed, 2020,95(3):513–520
Abd El-Wahab EW, Zein El-Abedin RA, Ahmed WM, et al. Validation of a Non-Laboratory Based Screening Tool for Predicting Non-Alcoholic Fatty Liver Disease in an Egyptian Setting. Am J Med Sci, 2020,360(6):662–677
Guo K, Xu S, Zeng Z. “Liver–gut” axis: A target of traditional Chinese medicine for the treatment of nonalcoholic fatty liver disease. Front Endocrinol, 2022,13:1050709
Sun CC, Zhang CY, Duan JX, et al. PTUPB ameliorates high-fat diet-induced nonalcoholic fatty liver disease by inhibiting NLRP3 inflammasome activation in mice. Biochem Biophys Res Commun, 2020,523(4):1020–1026
Lan Y, Wu S, Wang Y, et al. Association between blood copper and nonalcoholic fatty liver disease according to sex. Clin Nutr, 2021,40(4):2045–2052
Bórquez JC, Díaz-Castro F, Pino-de La Fuente F, et al. Mitofusin-2 induced by exercise modifies lipid droplet-mitochondria communication, promoting fatty acid oxidation in male mice with NAFLD. Metabolism, 2024,152:155765
You Y, Liang W. SIRT1 and SIRT6: The role in aging-related diseases. Biochim Biophys Acta Mol Basis Dis, 2023,1869(7):166815
Yamamoto H, Schoonjans K, Auwerx J. Sirtuin functions in health and disease. Mol Endocrinol, 2007,21(8):1745–1755
Zeng Y, Hua YQ, Wang W, et al. Modulation of SIRT1-mediated signaling cascades in the liver contributes to the amelioration of nonalcoholic steatohepatitis in high fat fed middle-aged LDL receptor knockout mice by dihydromyricetin. Biochem Pharmacol, 2020,175:113927
Mihanfar A, Akbarzadeh M, Ghazizadeh Darband S, et al. SIRT1: a promising therapeutic target in type 2 diabetes mellitus. Arch Physiol Biochem, 2024,130(1):13–28
Shen S, Shen M, Kuang L, et al. SIRT1/SREBPs-mediated regulation of lipid metabolism. Pharmacol Res, 2024,199:107037
Ren R, He Y, Ding D, et al. Aging exaggerates acute-on-chronic alcohol-induced liver injury in mice and humans by inhibiting neutrophilic sirtuin 1-C/EBPα-miRNA-223 axis. Hepatology, 2022,75(3):646–660
Zhang BB, Zhou G, Li C. AMPK: An Emerging Drug Target for Diabetes and the Metabolic Syndrome. Cell Metab, 2009,9(5):407–416
Liu J, Chen D, Liu P, et al. Discovery, synthesis, and structure–activity relationships of 20(S)-protopanaxadiol (PPD) derivatives as a novel class of AMPKα2β1γ1 activators. Eur J Med Chem, 2014,79:340–349
Cantó C, Gerhart Hines Z, Feige JN, et al. AMPK regulates energy expenditure by modulating NAD+ metabolism and SIRT1 activity. Nature, 2009,458(7241):1056–1060
Zhang D, Ding H, Liu C, et al. Circulating exosome-mediated AMPKα–SIRT1 pathway regulates lipid metabolism disorders in calf hepatocytes. Vet Res, 2024,169:105177
Yin X, Liu Z, Wang J. Tetrahydropalmatine ameliorates hepatic steatosis in nonalcoholic fatty liver disease by switching lipid metabolism via AMPK-SREBP-1c-Sirt1 signaling axis. Phytomedicine, 2023,119:155005
Zhou NN, Wang T, Lin YX, et al. Uridine alleviates high-carbohydrate diet-induced metabolic syndromes by activating sirt1/AMPK signaling pathway and promoting glycogen synthesis in Nile tilapia (Oreochromis niloticus). Ani Nutr, 2023,14:56–66
Fang K, Wu F, Chen G, et al. Diosgenin ameliorates palmitic acid-induced lipid accumulation via AMPK/ACC/CPT-1A and SREBP-1c/FAS signaling pathways in LO2 cells. BMC Complem Altern M, 2019,19(1):255
Xu A, Wang L, Luo M, et al. Overexpression of salusin-β downregulates adipoR1 expression to prevent fatty acid oxidation in HepG2 cells. Mol Med Rep, 2024,29(2):18
Liou CJ, Wei CH, Chen YL, et al. Fisetin Protects Against Hepatic Steatosis Through Regulation of the Sirt1/AMPK and Fatty Acid β-Oxidation Signaling Pathway in High-Fat Diet-Induced Obese Mice. Cell Physiol Biochem, 2018,49(5):1870–1884
Wang X, Liang F, Dai Z, et al. Combination of Coptis chinensis polysaccharides and berberine ameliorates ulcerative colitis by regulating gut microbiota and activating AhR/IL-22 pathway. J Ethnopharmacol, 2024,318:117050
Di Pierro F, Sultana R, Eusaph AZ, et al. Effect of Berberine Phytosome on reproductive, dermatologic, and metabolic characteristics in women with polycystic ovary syndrome: a controlled, randomized, multicentric, open-label clinical trial. Front Pharmacol, 2023,14:1269605
Gasmi A, Asghar F, Zafar S, et al. Berberine: Pharmacological Features in Health, Disease and Aging. Curr Med Chem, 2024,31(10):1214–1234
Tang Y, Gao Y, Nie K, et al. Jiao-tai-wan and its effective component-berberine improve diabetes and depressive disorder through the cAMP/PKA/CREB signaling pathway. J Ethnopharmacol, 2024,324:117829
Cai Y, Yang Q, Yu Y, et al. Efficacy and underlying mechanisms of berberine against lipid metabolic diseases: a review. Front Pharmacol, 2023,15:14:1283784
Tang J, Feng Y, Tsao S, et al. Berberine and Coptidis Rhizoma as novel antineoplastic agents: A review of traditional use and biomedical investigations. J Ethnopharmacol, 2009,126(1):5–17
Devarajan N, Nathan J, Mathangi R, et al. Pharmacotherapeutic values of berberine: A Chinese herbal medicine for the human cancer management. J Biochem Mol Toxic, 2023,37(3):e23278
Yan HM, Xia MF, Wang Y, et al. Efficacy of Berberine in Patients with Non-Alcoholic Fatty Liver Disease. PLoS One, 2015,10(8):e0134172
Nejati L, Movahedi A, Salari GR, et al. The Effect of Berberine on Lipid Profile, Liver Enzymes, and Fasting Blood Glucose in Patients with Nonalcoholic Fatty Liver Disease (NAFLD): A Randomized Controlled Trial. MJIRI, 2022,36(1):294–301
Koperska A, Wesolek A, Moszak M, et al. Berberine in Non-Alcoholic Fatty Liver Disease-A Review. Nutrients, 2022,14(17):3459
Dai Y, Zhu W, Zhou J, et al. The combination of berberine and evodiamine ameliorates high-fat diet-induced nonalcoholic fatty liver disease associated with modulation of gut microbiota in rats. Braz J Med Biol Res, 2022,55:e12096
Rafiei H, Yeung M, Kowalski S, et al. Development of a novel human triculture model of nonalcoholic fatty liver disease and identification of berberine as ameliorating steatosis, oxidative stress and fibrosis. Front Pharmacol, 2023,14:12343000
Utami AR, Maksum IP, Deawati Y. Berberine and Its Study as an Antidiabetic Compound. Biology, 2023,12(7):973
Zhang H, Zhao C, Cao G, et al. Berberine modulates amyloid-β peptide generation by activating AMP-activated protein kinase. Neuropharmacology, 2017,125:408–417
Wang P, Li R, Li Y, et al. Berberine alleviates nonalcoholic hepatic steatosis partially by promoting SIRT1 deacetylation of CPT1A in mice. Gastroenterol Rep (Oxf), 2023,11:goad032
Liu X, Wang K, Zhou J, et al. Metformin and Berberine suppress glycogenolysis by inhibiting glycogen phosphorylase and stabilizing the molecular structure of glycogen in db/db mice. Carbohydr Polym, 2020,243:116435
Brunt EM. Pathology of nonalcoholic fatty liver disease. Nat Rev Gastroenterol Hepatol, 2010,7(4):195–203
Klaunig JE, Li X, Wang Z. Role of xenobiotics in the induction and progression of fatty liver disease. Toxicol Res, 2018,7(4):664–680
Pizzino G, Irrera N, Cucinotta M, et al. Oxidative Stress: Harms and Benefits for Human Health. Oxid Med Cell Longev, 2017,2017:8416763
Friedman SL, Neuschwander-Tetri BA, Rinella M, et al. Mechanisms of NAFLD development and therapeutic strategies. Nat Med, 2018,24(7):908–922
Chen H, Zhou W, Ruan Y, et al. Reversal of angiotensin ll-induced β-cell dedifferentiation via inhibition of NF-κb signaling. Mol Med, 2018,24(1):43
Zhang W, Xu JH, Yu T, et al. Effects of berberine and metformin on intestinal inflammation and gut microbiome composition in db/db mice. Biomed Pharmacother, 2019,118:109131
Ciupińska-Kajor M, Hartleb M, Kajor M, et al. Hepatic angiogenesis and fibrosis are common features in morbidly obese patients. Hepatol Int, 2013,7(1):233–240
Ramalho RM, Cortez-Pinto H, Castro RE, et al. Apoptosis and Bcl-2 expression in the livers of patients with steatohepatitis. Eur J Gastroenterol Hepatol, 2006,18(1):21–29
Panasiuk A, Dzieciol J, Panasiuk B, et al. Expression of p53, Bax and Bcl-2 proteins in hepatocytes in nonalcoholic fatty liver disease. World J Gastroentero, 2006,12(38):5
Wu QJ, Zhang TN, Chen HH, et al. The sirtuin family in health and disease. Signal Transduct Target Ther, 2022,7(1):402
Meng D, Zhang F, Yu W, et al. Biological Role and Related Natural Products of SIRT1 in Nonalcoholic Fatty Liver. Diabet Metab Synd Ob, 2023,16:4043–4064
Yin H, Hu M, Liang X, et al. Deletion of SIRT1 From Hepatocytes in Mice Disrupts Lipin-1 Signaling and Aggravates Alcoholic Fatty Liver. Gastroenterology, 2014,146(3):801–811
Keshavjee B, Lambelet V, Coppola H, et al. Stress-Induced Premature Senescence Related to Oxidative Stress in the Developmental Programming of Nonalcoholic Fatty Liver Disease in a Rat Model of Intrauterine Growth Restriction. Antioxidants (Basel), 2022,11(9):1695
Lawrence T. The Nuclear Factor NF-κB Pathway in Inflammation. Cold Spring Harb Perspect Biol, 2009,1(6):a001651
Yeung F, Hoberg JE, Ramsey CS, et al. Modulation of NF-κB-dependent transcription and cell survival by the SIRT1 deacetylase. EMBO J, 2004,23(12):2369–2380
Rada P, Pardo V, Mobasher MA, et al. SIRT1 Controls Acetaminophen Hepatotoxicity by Modulating Inflammation and Oxidative Stress. Antioxid Redox Signal, 2017,28(13):1187–1208
Xu G, Zhao J, Liu H, et al. Melatonin Inhibits Apoptosis and Oxidative Stress of Mouse Leydig Cells via a SIRT1-Dependent Mechanism. Molecules, 2019,24(17):3084
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
This work was supported by grants from the National Natural Science Foundation of China (No. 81803799) and Hubei Province Natural Science Foundation of China (No. 2022CFB092).
Rights and permissions
About this article
Cite this article
Chen, C., Liu, Xc. & Deng, B. Protective Effects of Berberine on Nonalcoholic Fatty Liver Disease in db/db Mice via AMPK/SIRT1 Pathway Activation. CURR MED SCI (2024). https://doi.org/10.1007/s11596-024-2914-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11596-024-2914-y