Abstract
Objective
Kidney renal clear cell carcinoma (KIRC) is a common renal malignancy that has a poor prognosis. As a member of the F box family, cyclin F (CCNF) plays an important regulatory role in normal tissues and tumors. However, the underlying mechanism by which CCNF promotes KIRC proliferation still remains unclear.
Methods
Bioinformatics methods were used to analyze The Cancer Genome Atlas (TCGA) database to obtain gene expression and clinical prognosis data. The CCK8 assay, EdU assay, and xenograft assay were used to detect cell proliferation. The cell senescence and potential mechanism were assessed by SA-β-gal staining, Western blotting, as well as ELISA.
Results
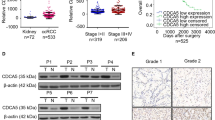
Our data showed that CCNF was highly expressed in KIRC patients. Meanwhile, downregulation of CCNF inhibited cell proliferation in vivo and in vitro. Further studies showed that the reduction of CCNF promoted cell senescence by decreasing cyclin-dependent kinase 1 (CDK1), increasing the proinflammatory factors interleukin (IL)-6 and IL-8, and then enhancing the expression of p21 and p53.
Conclusion
We propose that the high expression of CCNF in KIRC may play a key role in tumorigenesis by regulating cell senescence. Therefore, CCNF shows promise as a new biomarker to predict the clinical prognosis of KIRC patients and as an effective therapeutic target.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Padala SA, Barsouk A, Thandra KC, et al. Epidemiology of Renal Cell Carcinoma. World J Oncol, 2022, 11(3): 79–87
Hsieh JJ, Purdue MP, Signoretti S, et al. Renal Cell Carcinoma. Nat Rev Dis Primers, 2017,3:17009
Miller KD, Nogueira L, Mariotto AB, et al. Cancer Treatment and Survivorship Statistics, 2019. CA Cancer J Clin, 2019,69(5):363–385
Motzer RJ, Jonasch E, Agarwal N, et al. Kidney Cancer, Version 3.2015. J Natl Compr Canc Netw, 2015,13(2): 151–159
D’Angiolella V, Donato V, Vijayakumar S, et al. Scf(Cyclin F) Controls Centrosome Homeostasis and Mitotic Fidelity through Cp110 Degradation. Nature, 2010,466(7302):138–142
Yang ZL, Yang H, Tuo T, et al. Increased Expression of Cyclin F in Liver Cancer Predicts Poor Prognosis: A Study Based on Tcga Database. Medicine (Baltimore), 2021,100(31):e26623–e23
D’Angiolella V, Donato V, Forrester FM, et al. Cyclin F-Mediated Degradation of Ribonucleotide Reductase M2 Controls Genome Integrity and DNA Repair. Cell, 2012,149(5):1023–1034
Galper J, Rayner SL, Hogan AL, et al. Cyclin F: A Component of an E3 Ubiquitin Ligase Complex with Roles in Neurodegeneration and Cancer. Int J Biochem Cell Biol, 2017,89:216–220
Chang SC, Hung CS, Zhang BX, et al. A Novel Signature of Ccnf-Associated E3 Ligases Collaborate and Counter Each Other in Breast Cancer. Cancers (Basel), 2021,13(12):2873
Fu J, Qiu H, Cai M, et al. Low Cyclin F Expression in Hepatocellular Carcinoma Associates with Poor Differentiation and Unfavorable Prognosis. Cancer Sci, 2013,104(4):508–515
Xia S, Lin Y, Lin J, et al. Increased Expression of Ticrr Predicts Poor Clinical Outcomes: A Potential Therapeutic Target for Papillary Renal Cell Carcinoma. Front Genet, 2020,11:605378
Subramanian A, Tamayo P, Mootha VK, et al. Gene Set Enrichment Analysis: A Knowledge-Based Approach for Interpreting Genome-Wide Expression Profiles. Proc Natl Acad Sci USA, 2005,102(43):15545–15550
Yu G, Wang LG, Han Y, et al. Clusterprofiler: An R Package for Comparing Biological Themes among Gene Clusters. OMICS, 2012,16(5):284–287
Szklarczyk D, Gable AL, Lyon D, et al. String V11: Protein-Protein Association Networks with Increased Coverage, Supporting Functional Discovery in Genome-Wide Experimental Datasets. Nucleic Acids Res, 2019,47(D1):D607–D613
Liu J, Lichtenberg T, Hoadley KA, et al. An Integrated Tcga Pan-Cancer Clinical Data Resource to Drive High-Quality Survival Outcome Analytics. Cell, 2018,173(2):400–416.e11
Bai C, Richman R, Elledge SJ. Human Cyclin F. EMBO J, 1994,13(24):6087–6098
Li Y, Guo H, Wang Z, et al. Cyclin F and Kif20a, Foxm1 Target Genes, Increase Proliferation and Invasion of Ovarian Cancer Cells. Exp Cell Res, 2020,395(2):112212
Comisso E, Scarola M, Rosso M, et al. Oct4 Controls Mitotic Stability and Inactivates the Rb Tumor Suppressor Pathway to Enhance Ovarian Cancer Aggressiveness. Oncogene, 2017,36(30):4253–4266
Liao H, Ji F, Ying S. Cdk1: Beyond Cell Cycle Regulation. Aging (Albany NY), 2017,9(12):2465–2466
Bae T, Weon KY, Lee JW, et al. Restoration of Paclitaxel Resistance by Cdk1 Intervention in Drug-Resistant Ovarian Cancer. Carcinogenesis, 2015,36(12):1561–1571
Müllers E, Silva Cascales H, Burdova K, et al. Residual Cdk1/2 Activity after DNA Damage Promotes Senescence. Aging Cell, 2017,16(3):575–584
Riessland M, Kolisnyk B, Kim TW, et al. Loss of Satb1 Induces P21-Dependent Cellular Senescence in Post-Mitotic Dopaminergic Neurons. Cell Stem Cell, 2019,25(4):514–530–e8
Author information
Authors and Affiliations
Corresponding author
Additional information
Conflict of Interest Statement
The authors declare that they have no conflicts of interest regarding the publication of this paper.
This study was supported by the National Natural Science Foundation of China (No. 81874148 and No. 82203142).
Rights and permissions
About this article
Cite this article
Li, X., Li, Yj., Wang, Mj. et al. Inhibition of Cyclin F Promotes Cellular Senescence through Cyclin-dependent Kinase 1-mediated Cell Cycle Regulation. CURR MED SCI 43, 246–254 (2023). https://doi.org/10.1007/s11596-022-2692-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-022-2692-3