Abstract
Objective
Previous study suggested that estradiol (E2) plays an important role in otolith shedding by regulating the expression of otoconin 90 (OC90). The purpose of this article is to provide further data on the effect and mechanism of E2 on the morphology of otolith.
Methods
The rats receiving bilateral ovariectomy (OVX) were used as animal models. Co-immunoprecipitation was used to observe the relationship between estrogen receptor (ER) and estrogen-related receptor α (ERRα). The morphology of otolith was observed under the scanning electron microscopy. Western blotting and qPCR were used for quantitative analysis of the roles of ER and ERRα in regulating OC90 expression.
Results
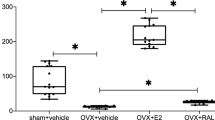
The looser otoliths were observed in rats receiving bilateral OVX, which could be reversed by supplementation with E2. The level of ERRα was decreased in bilateral OVX rats. ER and ERRα interacted with each other on the regulation of the expression of OC90.
Conclusion
Our results suggest ER and ERRα are both important downstream receptors involved in regulating OC90 expression in utricles of rats, and ERRα probably functions by interacting with ER. This provides evidence for the mechanism of otolith shedding. And it may be significant for future studies of targeted prevention and therapies for benign paroxysmal positional vertigo.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Vibert D, Sans A, Kompis M, et al. Ultrastructural changes in otoconia of osteoporotic rats. Audiol Neurootol, 2008,13(5):293–301
Yang H, Gu H, Sun W, et al. Estradiol deficiency is a risk factor for idiopathic benign paroxysmal positional vertigo in postmenopausal female patients. Laryngoscope, 2018,128(4):948–953
Wang Y, Kowalski PE, Thalmann I, et al. Otoconin-90, the mammalian otoconial matrix protein, contains two domains of homology to secretory phospholipase A2. Proc Nat Acad Sci USA, 1998,95(26):15 345–15 350
Verpy E, Leibovici M, Petit C. Characterization of otoconin-95, the major protein of murine otoconia, provides insights into the formation of these inner ear biominerals. Proc Nat Acad Sci USA, 1999,96(2):529–534
Heldring N, Pike A, Andersson S, et al. Estrogen receptors: how do they signal and what are their targets. Physiol Rev, 2007,87(3):905–931
Simonoska R, Stenberg A, Masironi B, et al. Estrogen receptors in the inner ear during different stages of pregnancy and development in the rat. Acta Otolaryngol, 2009,129(11):1175–1181
Motohashi R, Takumida M, Shimizu A, et al. Effects of age and sex on the expression of estrogen receptor alpha and beta in the mouse inner ear. Acta Otolaryngol, 2010,130(2):204–214
Roos W, Oeze L, Löser R, et al. Antiestrogenic action of 3-hydroxytamoxifen in the human breast cancer cell line MCF-7. J Natl Cancer Inst, 1983,71(1):55–59
Henley DV, Korach KS. Physiological effects and mechanisms of action of endocrine disrupting chemicals that alter estrogen signaling. Hormones (Athens), 2010,9(3):191–205
Bonnelye E, Merdad L, Kung V, et al. The orphan nuclear estrogen receptor-related receptor alpha (ERRalpha) is expressed throughout osteoblast differentiation and regulates bone formation in vitro. J Cell Biol, 2001,153(5):971–984
Horard B, Vanacker JM. Estrogen receptor-related receptors: orphan receptors desperately seeking a ligand. Journal of molecular endocrinology. J Mol Endocrinol, 2003,31(3):349–357
Bonnelye E, Vanacker JM, Dittmar T, et al. The ERR-1 orphan receptor is a transcriptional activator expressed during bone development. Mol Endocrinol, 1997,11(7):905–916
Landis WJ. The strength of a calcified tissue depends in part on the molecular structure and organization of its constituent mineral crystals in their organic matrix. Bone, 1995,16(5):533–544
Lu X, Peng L, Lv M, et al. Recent advance in the design of small molecular modulators of estrogen-related receptors. Curr Pharm Des, 2012,18(23):3421–3431
Wei W, Schwaid AG, Wang X, et al. Ligand Activation of ERRα by Cholesterol Mediates Statin and Bisphosphonate Effects. Cell Metab, 2016,23(3):479–491
Zhang J, Peng Z, Yang M, et al. Observation of the morphology and calcium content of vestibular otoconia in rats after simulated weightlessness. Acta Otolaryngol, 2005,125(10):1039–1042
Hamann KF. Benign paroxysmal positioning vertigo: a disease explainable by inner ear mechanics. ORL J Otorhinolaryngol Relat Spec, 2006,68(6):329–333
Yang H, Zhao X, Xu Y, et al. Matrix recruitment and calcium sequestration for spatial specific otoconia development. PloS One, 2016(5):e20498
Zhao X, Yang H, Yamoah EN, et al. Gene targeting reveals the role of Oc90 as the essential organizer of the otoconial organic matrix. Dev Biol, 2007,304(2):508–524
Acknowledgments
Thanks to Professor Ding Dalian for his help in teaching the extraction method of inner ear tissue.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
All authors declare no conflicts of interest.
Additional information
This work was supported by grants from Scientific Research Plan Project of Science and Technology Commission of Shanghai Municipality (No. 18ZR1439200) and Talents Training Program of Pudong Hospital affiliated to Fudan University (No. PX201705).
Rights and permissions
About this article
Cite this article
Feng, My., Gu, Hh., Tian, Q. et al. Molecular Mediators of Estrogen Reduction-induced Otolith Shedding. CURR MED SCI 41, 667–672 (2021). https://doi.org/10.1007/s11596-021-2421-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-021-2421-3