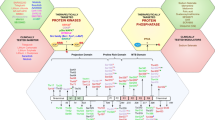
Summary
Protein phosphatase 2A (PP2A) is a major serine/threonine phosphatase which participates in the regulation of multiple cellular processes. As a confirmed tumor suppressor, PP2A activity is downregulated in tumors and its re-activation can induce apoptosis of cancer cells. In the brains of Alzheimer’s disease (AD) patients, decreased PP2A activity also plays a key role in promoting tau hyperphosphorylation and Aβ generation. In this review, we discussed compounds aiming at modulating PP2A activity in the treatment of cancer or AD. The upstream factors that inactivate PP2A in diseases have not been fully elucidated and further studies are needed. It will help for the refinement and development of novel and clinically tractable PP2A-targeted compounds or therapies for the treatment of tumor and AD.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Change history
26 April 2020
The article ���Protein Phosphatase 2A as a Drug Target in the Treatment of Cancer and Alzheimer���s Disease���, written by Hui WEI, Hui-liang ZHANG, Jia-zhao XIE, Dong-li MENG, Xiao-chuan WANG, Dan KE, Ji ZENG, Rong LIU, was originally published electronically on the publisher���s internet portal on 13 March 2020 without open access. With the author(s)��� decision to opt for Open Choice the copyright of the article changed to �� The Author(s) 2020 and the article is forthwith distributed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
The original article has been corrected.
Corresponding authors: Dan KE, E-mail: kedan@hust.edu.cn; Ji ZENG, E-mail: whzjmicro@163.com
References
Chen MJ, Dixon JE, Manning G. Genomics and evolution of protein phosphatases. Sci Signal, 2017,10 (474). pii: eaag1796.
Ingebritsen TS, Cohen P. The protein phosphatases involved in cellular regulation. 1. Classification and substrate specificities. Eur J Biochem, 1983,132(2):255–261
Shi Y. Serine/Threonine Phosphatases: Mechanism through Structure. Cell, 2009,139(3):468–484
Voronkov M, Braithwaite SP, Stock JB. Phosphoprotein phosphatase 2A: a novel druggable target for Alzheimer’s disease. Future Med Chem, 2011,3(7):821–833
Sangodkar J, Farrington CC, McClinch K, et al. All roads lead to PP2A: exploiting the therapeutic potential of this phosphatase. FEBS J, 2016,283(6):1004–1024
Janssens V, Goris J. Protein phosphatase 2A: a highly regulated family of serine/threonine phosphatases implicated in cell growth and signalling. Biochem J, 2001,353(Pt 3):417–439
Cho US, Xu W. Crystal structure of a protein phosphatase 2A heterotrimeric holoenzyme. Nature, 2007,445(7123):53–57
Liu R, Wang JZ. Protein phosphatase 2A in Alzheimer’s disease. Pathophysiology, 2009,16(4):273–277
Janssens V, Goris J, Van Hoof C. PP2A: the expected tumor suppressor. Curr Opin Genet Dev, 2005,15(1):34–41
Liu R, Tian Q. Protein phosphatase 2A, a key player in Alzheimer’s disease. Front Med China, 2009,3(1):8–12
Neviani P, Santhanam R, Trotta R, et al. The tumor suppressor PP2A is functionally inactivated in blast crisis CML through the inhibitory activity of the BCR/ABL-regulated SET protein. Cancer Cell, 2005,8(5):355–368
Kalev P, Sablina AA. Protein phosphatase 2A as a potential target for anticancer therapy. Anticancer Agents Med Chem, 2011,11(1):38–46
Perrotti D, Neviani P. Protein phosphatase 2A: a target for anticancer therapy. Lancet Oncol, 2013,14(6):e229–e238
Longin S, Zwaenepoel K, Louis JV, et al. Selection of protein phosphatase 2A regulatory subunits is mediated by the C terminus of the catalytic Subunit. J Biol Chem, 2007,282(37):26971–26980
Jackson JB, Pallas DC. Circumventing cellular control of PP2A by methylation promotes transformation in an Akt-dependent manner. Neoplasia, 2012,14(7):585–599
Junttila MR, Puustinen P, Niemela M, et al. CIP2A inhibits PP2A in human malignancies. Cell, 2007,130(1):51–62
Li M, Guo H, Damuni Z. Purification and characterization of two potent heat-stable protein inhibitors of protein phosphatase 2A from bovine kidney. Biochemistry, 1995,34(6):1988–1996
Zhang J, Yang PL, Gray NS. Targeting cancer with small molecule kinase inhibitors. Nat Rev Cancer, 2009,9(1):28–39
Kim SH, Seo MS, Jeon WJ, et al. Haloperidol regulates the phosphorylation level of the MEK-ERK-p90RSK signal pathway via protein phosphatase 2A in the rat frontal cortex. Int J Neuropsychopharmacol, 2008,11(4):509–517
Gutierrez A, Pan L, Groen RW, et al. Phenothiazines induce PP2A-mediated apoptosis in T cell acute lymphoblastic leukemia. J Clin Invest, 2014,124(2):644–655
Sangodkar J, Perl A, Tohme R, et al. Activation of tumor suppressor protein PP2A inhibits KRAS-driven tumor growth. J Clin Invest, 2017,127(6):2081–2090
Li M, Makkinje A, Damuni Z. The myeloid leukemia-associated protein SET is a potent inhibitor of protein phosphatase 2A. J Biol Chem, 1996,271(19):11 059–11 062
Mukhopadhyay A, Saddoughi SA, Song P, et al. Direct interaction between the inhibitor 2 and ceramide via sphingolipid-protein binding is involved in the regulation of protein phosphatase 2A activity and signaling. FASEB J, 2009,23(3):751–763
Saddoughi SA, Gencer S, Peterson YK, et al. Sphingosine analogue drug FTY720 targets I2PP2A/SET and mediates lung tumour suppression via activation of PP2A-RIPK1-dependent necroptosis. EMBO Mol Med, 2013,5(1):105–121
Christensen DJ, Chen Y, Oddo J, et al. SET oncoprotein overexpression in B-cell chronic lymphocytic leukemia and non-Hodgkin lymphoma: a predictor of aggressive disease and a new treatment target. Blood, 2011,118(15):4150–4158
Janghorban M, Farrell AS, Allen-Petersen BL, et al. Targeting c-MYC by antagonizing PP2A inhibitors in breast cancer. Proc Natl Acad Sci, 2014,111(25):9157–9162
Agarwal A, MacKenzie RJ, Pippa R, et al. Antagonism of SET Using OP449 Enhances the Efficacy of Tyrosine Kinase Inhibitors and Overcomes Drug Resistance in Myeloid Leukemia. Clin Cancer Res, 2014,20(8):2092–2103
Liu Z, Ma L, Wen ZS, et al. Cancerous inhibitor of PP2A is targeted by natural compound celastrol for degradation in non-small-cell lung cancer. Carcinogenesis, 2014,35(4):905–914
Come C, Laine A, Chanrion M, et al. CIP2A is associated with human breast cancer aggressivity. Clin Cancer Res, 2009,15(16):5092–5100
Khanna A, Bockelman C, Hemmes A, et al. MYC-dependent regulation and prognostic role of CIP2A in gastric cancer. J Natl Cancer Inst, 2009,101(11):793–805
Corson TW, Crews CM. Molecular understanding and modern application of traditional medicines: triumphs and trials. Cell, 2007,130(5):769–774
Shrivastava S, Jeengar MK, Reddy VS, et al. Anticancer effect of celastrol on human triple negative breast cancer: possible involvement of oxidative stress, mitochondrial dysfunction, apoptosis and PI3K/Akt pathways. Exp Mol Pathol, 2015,98(3):313–327
Mou H, Zheng Y, Zhao P, et al. Celastrol induces apoptosis in non-small-cell lung cancer A549 cells through activation of mitochondria- and Fas/FasL-mediated pathways. Toxicol In Vitro, 2011,25(5):1027–1032
Yu X, Zhou X, Fu C, et al. Celastrol induces apoptosis of human osteosarcoma cells via the mitochondrial apoptotic pathway. Oncol Rep, 2015,34(3):1129–1136
Lee JH, Won YS, Park KH, et al. Celastrol inhibits growth and induces apoptotic cell death in melanoma cells via the activation ROS-dependent mitochondrial pathway and the suppression of PI3K/AKT signaling. Apoptosis, 2012,17(12):1275–1286
Liu Z, Ma L, Wen Z, et al. Ethoxysanguinarine Induces Inhibitory Effects and Downregulates CIP2A in Lung Cancer Cells. ACS Med Chem Lett, 2014,5(2):113–118
Miao F, Yang X, Zhou L, et al. Structural modification of sanguinarine and chelerythrine and their antibacterial activity. Nat Prod Res, 2011,25(9):863–875
Homma T, Fujii J. An SOD1 deficiency aggravates proteasome inhibitor bortezomib-induced testicular damage in mice. Biochim Biophys Acta Gen Subj, 2019,1863(6):1108–1115
Yu HC, Hou DR, Liu CY, et al. Cancerous inhibitor of protein phosphatase 2A mediates bortezomib-induced autophagy in hepatocellular carcinoma independent of proteasome. PLoS One, 2013,8(2):e55705
Tseng LM, Liu CY, Chang KC, et al. CIP2A is a target of bortezomib in human triple negative breast cancer cells. Breast Cancer Res, 2012,14(2):R68
Umemura S, Yoshida S, Ohta Y, et al. Increased phosphorylation of Akt in triple-negative breast cancers. Cancer Sci, 2007,98(12):1889–1892
Chao TT, Wang CY, Lai CC, et al. TD-19, an Erlotinib Derivative, Induces Epidermal Growth Factor Receptor Wild-Type Nonsmall-Cell Lung Cancer Apoptosis through CIP2A-Mediated Pathway. J Pharmacol Exp Ther, 2014,351(2):352–358
Yu H, Hung M, Chen Y, et al. Erlotinib derivative inhibits hepatocellular carcinoma by targeting CIP2A to reactivate protein phosphatase 2A. Cell Death Dis, 2014,5(7):e1359.
Connor CM, Perl A, Leonard D, et al. Therapeutic targeting of PP2A. Int J Biochem Cell Biol, 2018,96:182–193
Chen K, Pao K, Su J, et al. Development of erlotinib derivatives as CIP2A-ablating agents independent of EGFR activity. Bioorg Med Chem, 2012,20(20):6144–6153
Yu HC, Chen HJ, Chang YL, et al. Inhibition of CIP2A determines erlotinib-induced apoptosis in hepatocellular carcinoma. Biochem Pharmacol, 2013,85(3):356–366
Liu C, Huang T, Huang C, et al. EGFR-independent Elk1/CIP2A signalling mediates apoptotic effect of an erlotinib derivative TD52 in triple-negative breast cancer cells. Eur J Cancer, 2017,72:112–123
Chung V, Mansfield AS, Braiteh F, et al. Safety, Tolerability, and Preliminary Activity of LB-100, an Inhibitor of Protein Phosphatase 2A, in Patients with Relapsed Solid Tumors: An Open-Label, Dose Escalation, First-in-Human, Phase I Trial. Clin Cancer Res, 2017,23(13):3277–3284
Wang S, Xie W, Wang D, et al. Discovery of a small molecule targeting SET-PP2A interaction to overcome BCR-ABLT315I mutation of chronic myeloid leukemia. Oncotarget, 2015,6(14):12128–12140
Glenner GG, Wong CW. Alzheimer’s disease: initial report of the purification and characterization of a novel cerebrovascular amyloid protein. Biochem Biophys Res Commun, 1984,120(3):885–890
Grundke-Iqbal I, Iqbal K, Quinlan M, et al. Microtubule-associated protein tau. A component of Alzheimer paired helical filaments. J Biol Chem, 1986,261(13):6084–6089
Iqbal K, Grundke-Iqbal I, Zaidi T, et al. Defective brain microtubule assembly in Alzheimer’s disease. Lancet, 1986,2(8504):421–426
Wang J, Xia Y, Grundke-Iqbal I, et al. Abnormal Hyperphosphorylation of Tau: Sites, Regulation, and Molecular Mechanism of Neurofibrillary Degeneration. J Alzheimers Dis, 2012,33(s1):S123–S139
Grundke-Iqbal I, Iqbal K, Tung YC, et al. Abnormal phosphorylation of the microtubule-associated protein X (tau) in Alzheimer cytoskeletal pathology. Proc Natl Acad Sci USA, 1986,83(13):4913–4917
Gong CX, Shaikh S, Wang JZ, et al. Phosphatase activity toward abnormally phosphorylated tau: decrease in Alzheimer disease brain. J Neurochem, 1995,65(2):732–738
Tanimukai H, Kudo T, Tanaka T, et al. Novel therapeutic strategies for neurodegenerative disease. Psychogeriatrics, 2009,9(2):103–109
Cho JH, Johnson GV. Glycogen synthase kinase 3 beta induces caspase-cleaved tau aggregation in situ. J Biol Chem, 2004,279(52):54716–54723
Vogelsberg-Ragaglia V, Schuck T, Trojanowski JQ, et al. PP2A mRNA expression is quantitatively decreased in Alzheimer’s disease hippocampus. Exp Neurol, 2001,168(2):402–412
Sontag E, Luangpirom A, Hladik C, et al. Altered expression levels of the protein phosphatase 2A ABalphaC enzyme are associated with Alzheimer disease pathology. J Neuropathol Exp Neurol, 2004,63(4):287–301
Gong CX, Singh TJ, Grundke-Iqbal I, et al. Phosphoprotein phosphatase activities in Alzheimer disease brain. J Neurochem, 1993,61(3):921–927
Wang J, Tung YC, Wang Y, et al. Hyperphosphorylation and accumulation of neurofilament proteins in Alzheimer disease brain and in okadaic acid-treated SY5Y cells. FEBS Lett, 2001,507(1):81–87
Liu F, Grundke-Iqbal I, Iqbal K, et al. Contributions of protein phosphatases PP1, PP2A, PP2B and PP5 to the regulation of tau phosphorylation. Eur J Neurosci, 2005,22(8):1942–1950
Lee MS, Kao SC, Lemere CA, et al. APP processing is regulated by cytoplasmic phosphorylation. J Cell Biol, 2003,163(1):83–95
Colombo A, Bastone A, Ploia C, et al. JNK regulates APP cleavage and degradation in a model of Alzheimer’s disease. Neurobiol Dis, 2009,33(3):518–525
Braithwaite SP, Schmid RS, He DN, et al. Inhibition of c-Jun kinase provides neuroprotection in a model of Alzheimer’s disease. Neurobiol Dis, 2010,39(3):311–317
Dhanasekaran DN, Reddy EP. JNK signaling in apoptosis. Oncogene, 2008,27(48):6245–6251
Sontag E, Nunbhakdi-Craig V, Sontag JM, et al. Protein phosphatase 2A methyltransferase links homocysteine metabolism with tau and amyloid precursor protein regulation. J Neurosci, 2007,27(11):2751–2759
St-Cyr Giguère F, Attiori Essis S, Chagniel L, et al. The sphingosine-1-phosphate receptor 1 agonist SEW2871 reduces Tau-Ser262 phosphorylation in rat hippocampal slices. Brain Res, 2017,1658:51–59
Mizugishi K, Yamashita T, Olivera A, et al. Essential role for sphingosine kinases in neural and vascular development. Mol Cell Biol, 2005,25(24):11 113–11 121
Domise M, Didier S, Marinangeli C, et al. AMP-activated protein kinase modulates tau phosphorylation and tau pathology in vivo. Sci Rep, 2016,6:26758
Iijima K, Gatt A, Iijima-Ando K. Tau Ser262 phosphorylation is critical for Abeta42-induced tau toxicity in a transgenic Drosophila model of Alzheimer’s disease. Hum Mol Genet, 2010,19(15):2947–2957
Wang X, Xiong Y, Yang Y, et al. A novel tacrine-dihydropyridine hybrid (−) SCR1693 induces tau dephosphorylation and inhibits Aβ generation in cells. Eur J Pharmacol, 2015,754:134–139
Xia Y, Liu R, Chen R, et al. Novel Multipotent AChEI-CCB Attenuates Hyperhomocysteinemia-Induced Memory Deficits and Neuropathologies in Rats. J Alzheimers Dis, 2014,42(3):1029–1039
Miller LM, Wang Q, Telivala TP, et al. Synchrotron-based infrared and X-ray imaging shows focalized accumulation of Cu and Zn co-localized with beta-amyloid deposits in Alzheimer’s disease. J Struct Biol, 2006,155(1):30–37
Suh SW, Jensen KB, Jensen MS, et al. Histochemically-reactive zinc in amyloid plaques, angiopathy, and degenerating neurons of Alzheimer’s disease brains. Brain Res, 2000,852(2):274–278
Mezzaroba L, Alfieri DF, Colado SA, et al. The role of zinc, copper, manganese and iron in neurodegenerative diseases. Neurotoxicology, 2019,74:230–241
Xiong Y, Luo D, Wang X, et al. Zinc binds to and directly inhibits protein phosphatase 2A in vitro. Neurosci Bull, 2015,31(3):331–337
Xiong Y, Jing XP, Zhou XW, et al. Zinc induces protein phosphatase 2A inactivation and tau hyperphosphorylation through Src dependent PP2A (tyrosine 307) phosphorylation. Neurobiol Aging, 2013,34(3):745–756
Beard JL. Iron biology in immune function, muscle metabolism and neuronal functioning. J Nutr, 2001,131(2S-2):568S–579S, 580S
Tao Y, Wang Y, Rogers JT, et al. Perturbed iron distribution in Alzheimer’s disease serum, cerebrospinal fluid, and selected brain regions: a systematic review and meta-analysis. J Alzheimers Dis, 2014,42(2):679–690
Amit T, Avramovich-Tirosh Y, Youdim MB, et al. Targeting multiple Alzheimer’s disease etiologies with multimodal neuroprotective and neurorestorative iron chelators. FASEB J, 2008,22(5):1296–1305
Liu B, Moloney A, Meehan S, et al. Iron promotes the toxicity of amyloid beta peptide by impeding its ordered aggregation. J Biol Chem, 2011,286(6):4248–4256
Salissou M, Mahaman Y, Zhu F, et al. Methanolic extract of Tamarix Gallica attenuates hyperhomocysteinemia induced AD-like pathology and cognitive impairments in rats. Aging (Albany NY), 2018,10(11):3229–3248
Zhang Q, Xia Y, Luo H, et al. Codonopsis pilosula Polysaccharide Attenuates Tau Hyperphosphorylation and Cognitive Impairments in hTau Infected Mice. Front Mol Neurosci, 2018,11:437
Zeng K, Li M, Hu J, et al. Ginkgo biloba Extract EGb761 Attenuates Hyperhomocysteinemia-induced AD Like Tau Hyperphosphorylation and Cognitive Impairment in Rats. Curr Alzheimer Res, 2018,15(1):89–99
Mahaman Y, Huang F, Wu M, et al. Moringa Oleifera Alleviates Homocysteine-Induced Alzheimer’s Disease-Like Pathology and Cognitive Impairments. J Alzheimers Dis, 2018,63(3):1141–1159
Chai GS, Jiang X, Ni ZF, et al. Betaine attenuates Alzheimer-like pathological changes and memory deficits induced by homocysteine. J Neurochem, 2013,124(3):388–396
Zhang Y, Xie JZ, Xu XY, et al. Liraglutide Ameliorates Hyperhomocysteinemia-Induced Alzheimer-Like Pathology and Memory Deficits in Rats via Multi-molecular Targeting. Neurosci Bull, 2019,35(4):724–734
Zhou P, Chen Z, Zhao N, et al. Acetyl-L-carnitine attenuates homocysteine-induced Alzheimer-like histopathological and behavioral abnormalities. Rejuvenation Res, 2011,14(6):669–679
Zeng P, Shi Y, Wang XM, et al. Emodin Rescued Hyperhomocysteinemia-Induced Dementia and Alzheimer’s Disease-Like Features in Rats. Int J Neuropsychopharmacol, 2019,22(1):57–70
Wei W, Liu YH, Zhang CE, et al. Folate/vitamin-B12 prevents chronic hyperhomocysteinemia-induced tau hyperphosphorylation and memory deficits in aged rats. J Alzheimers Dis, 2011,27(3):639–650
Lee KW, Chen W, Junn E, et al. Enhanced phosphatase activity attenuates alpha-synucleinopathy in a mouse model. J Neurosci, 2011,31(19):6963–6971
Guenin S, Schwartz L, Morvan D, et al. PP2A activity is controlled by methylation and regulates oncoprotein expression in melanoma cells: a mechanism which participates in growth inhibition induced by chloroethylnitrosourea treatment. Int J Oncol, 2008,32(1):49–57
Wu Y, Song P, Xu J, et al. Activation of protein phosphatase 2A by palmitate inhibits AMP-activated protein kinase. J Biol Chem, 2007,282(13):9777–9788
Yang X, Yang Y, Fu Z, et al. Melatonin ameliorates Alzheimer-like pathological changes and spatial memory retention impairment induced by calyculin A. J Psychopharmacol, 2011,25(8):1118–1125
Cho DH, Choi YJ, Jo SA, et al. Troglitazone acutely inhibits protein synthesis in endothelial cells via a novel mechanismin volving protein phosphatase 2A-dependent p70 S6 kinase inhibition. Am J Physiol Cell Physiol, 2006,291(2):C317–326
Liu B, Arbogast LA. Progesterone decreases tyrosine hydroxylase phosphorylation state and increases protein phosphatase 2A activity in the stalk-median eminence on proestrous afternoon. J Endocrinol, 2010,204(2):209–219
Switzer CH, Ridnour LA, Cheng RY, et al. Dithiolethione compounds inhibit Akt signaling in human breast and lung cancer cells by increasing PP2A activity. Oncogene, 2009,28(43):3837–3846
Aceto N, Bertino P, Barbone D, et al. Taurolidine and oxidative stress: a rationale for local treatment of mesothelioma. Eur Respir J, 2009,34(6):1399–1407
Neviani P, Santhanam R, Trotta R, et al. The tumor suppressor PP2A is functionally inactivated in blast crisis CML through the inhibitory activity of the BCR/ABL-regulated SET protein. Cancer Cell, 2005,8(5):355–368
Perrotti D, Neviani P. Protein phosphatase 2A (PP2A), a drugable tumor suppressor in Ph1 (+) leukemias. Cancer Metastasis Rev, 2008,27(2):159–168
Nakashima I, Liu W, Akhand AA, et al. 4-hydroxynonenal triggers multistep signal transduction cascades for suppression of cellular functions. Mol Aspects Med, 2003,24(4–5):231–238
Liu W, Akhand AA, Takeda K, et al. Protein phosphatase 2A-linked and -unlinked caspase-dependent pathways for downregulation of Akt kinase triggered by 4-hydroxynonenal. Cell Death Differ, 2003,10(7):772–781
Kowluru A, Seavey SE, Rabaglia ME, et al. Carboxylmethylation of the catalytic subunit of protein phosphatase 2A in insulin-secreting cells: evidence for functional consequences on enzyme activity and insulin secretion. Endocrinology, 1996,137(6):2315–2323
Kitano K, Nam KY, Kimura S, et al. Sealing effects of (−)-epigallocatechin gallate on protein kinase C and protein phosphatase 2A. Biophys Chem, 1997,65(2–3):157–164
de Los Ríos C, Egea J, Marco-Contelles J, et al, Synthesis, inhibitory activity of cholinesterases, and neuroprotective profile of novel 1,8-naphthyridine derivatives. J Med Chem, 2010,53(14):5129–5143
Author information
Authors and Affiliations
Corresponding authors
Additional information
Conflict of Interest Statement
The authors have declared that no conflict of interest exists.
The original version of this article was revised due to a retrospective Open Access order
This study was supported by grants from National Natural Science Foundation of China (No. 31970964 and No. 31771189), Natural Science Foundation of Hubei Province, China (No. 2017CFA065) and Wuhan Health and Family Planning Commission (No. WX18C17).
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License http://creativecommons.org/licenses/by/4.0/, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wei, H., Zhang, Hl., Xie, Jz. et al. Protein Phosphatase 2A as a Drug Target in the Treatment of Cancer and Alzheimer’s Disease. CURR MED SCI 40, 1–8 (2020). https://doi.org/10.1007/s11596-020-2140-1
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-020-2140-1