Abstract
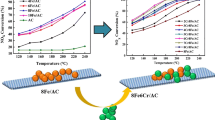
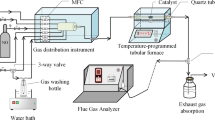
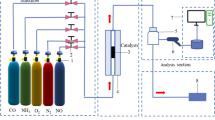
To study the modification mechanism of activated carbon (AC) by Fe and the low-temperature NH3-selective catalytic reduction (SCR) denitration mechanism of Fe/AC catalysts, Fe/AC catalysts were prepared using coconut shell AC activated by nitric acid as the support and iron oxide as the active component. The crystal structure, surface morphology, pore structure, functional groups and valence states of the active components of Fe/AC catalysts were characterised by X-ray diffraction, scanning electron microscopy, nitrogen adsorption and desorption, Fourier transform infrared spectroscopy and X-ray photoelectron spectroscopy, respectively. The effect of Fe loading and calcination temperature on the low-temperature denitration of NH3-SCR over Fe/AC catalysts was studied using NH3 as the reducing gas at low temperature (150 °C). The results show that the iron oxide on the Fe/AC catalyst is spherical and uniformly dispersed on the surface of AC, thereby improving the crystallisation performance and increasing the number of active sites and specific surface area on AC in contact with the reaction gas. Hence, a rapid NH3-SCR reaction was realised. When the roasting temperature remains constant, the iron oxide crystals formed by increasing the amount of loading can enter the AC pore structure and accumulate to form more micropores. When the roasting temperature is raised from 400 to 500 °C, the iron oxide is mainly transformed from α-Fe2O3 to γ-Fe2O3, which improves the iron oxide dispersion and increases its denitration active site, allowing gas adsorption. When the Fe loading amount is 10%, and the roasting temperature is 500 °C, the NO removal rate of the Fe/AC catalyst can reach 95%. According to the study, the low-temperature NH3-SCR mechanism of Fe/AC catalyst is proposed, in which the redox reaction between Fe2+ and Fe3+ will facilitate the formation of reactive oxygen vacancies, which increases the amount of oxygen adsorption on the surface, especially the increase in surface acid sites, and promotes and adsorbs more reaction gases (NH3, O2, NO). The transformation from the standard SCR reaction to the fast SCR reaction is accelerated.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Liu Y X, Zhang J, Pan J F, et al. Investigation on the Removal of NO from SO2-Containing Simulated Flue Gas by an Ultraviolet/Fenton-Like Reaction[J]. Energy Fuels, 2012, 26(9): 5 430–5 436
Song X Y, Ma X L, Ning G Q, et al. Pitch-Based Nitrogen-Doped Mesoporous Carbon for Flue Gas Desulfurization[J]. Ind. Eng. Chem. Res., 2017, 56(16): 4 743–4 749
Ye J H, Shang J, Li Q, et al. The Use of Vacuum Ultraviolet Irradiation to Oxidize SO2 and NOx for Simultaneous Desulfurization and Denitrification[J]. J. Hazard. Mater., 2014, 271: 89–97
Zhan S H, Zhang H, Zhang Y, et al. Efficient NH3-SCR Removal of NOx with Highly Ordered Mesoporous WO3(chi)-CeO2 at Low Temperatures[J]. Appl. Catal. B-Environ., 2017, 203: 199–209
Meng D M, Zhan W C, Guo Y, et al. A Highly Effective Catalyst of Sm-Mn Mixed Oxide for the Selective Catalytic Reduction of NOx with Ammonia: Effect of the Calcination Temperature[J]. J. Mol. Catal. A-Chem., 2016, 420: 272–281
Liu Y, Ning P, Li K, et al. Simultaneous Removal of NOx and SO2 by Low-Temperature Selective Catalytic Reduction over Modified Activated Carbon Catalysts[J]. Russ. J. Phys. Chem. A+, 2017, 91(3): 490–499
Nieto-Delgado C, Gutierrez-Martinez J, Rangel-Mendez J R. Modified Activated Carbon with Interconnected Fibrils of Iron-Oxyhydroxides using Mn2+ as Morphology Regulator, for a Superior Arsenic Removal from Water[J]. J. Environ. Sci., 2019, 76: 403–414
Jiang L J, Liu Q C, Zhao Q, et al. Promotional Effect of Ce on the SCR of NO with NH3 at Low Temperature over MnOx supported by Nitric Acid-Modified Activated Carbon[J]. Res. Chem. Intermediater., 2018, 44(3): 1 729–1 744
Nielsen L, Biggs M J, Skinner W, et al. The Effects of Activated Carbon Surface Features on the Reactive Adsorption of Carbamazepine and Sulfamethoxazole[J]. Carbon, 2014, 80: 419–432
Yang R, Huang H F, Chen Y J, et al. Performance of Cr-doped Vanadia/Titania Catalysts for Low-Temperature Selective Catalytic Reduction of NOx with NH3[J]. Chinese J. Catal., 2015, 36(8): 1 256–1 262
Yang J J, Qing M, Li W, et al. Effect of Nano V(2)O(5), Nano Fe(2)O(3) and Nano V(2)O(5)/Fe2O3 on Selective Catalytic Reduction of NO over a Modified AC Catalyst[J]. Int. J. Oil. Gas. Coal. T., 2016, 11(4): 387–396
Xu W T, Zhou J C, Li H, et al. Microwave-Assisted Catalytic Reduction of NO into N2 by Activated Carbon Supported Mn2O3 at Low Temperature under O2 Excess[J]. Fuel Process. Technol., 2014, 127: 1–6
Lei Z G, Han B, Yang K, et al. Influence of H2O on the Low-Temperature NH3-SCR of NO over V2O5/AC Catalyst: An Experimental and Modeling Study[J]. Chem. Eng. J., 2013, 215: 651–657
Chen Y, Liao Y F, Chen L, et al. Performance of Transition Metal (Cu, Fe and Co) Modified SCR Catalysts for Simultaneous Removal of NO and Volatile Organic Compounds (VOCs) from Coal-Fired Power Plant Flue Gas[J]. Fuel, 2021, 289: 119 849
Zhang X L, Lv S S, Zhang X C, et al. Improvement of the Activity and SO2 Tolerance of Sb-modified Mn/PG Catalysts for NH3-SCR at a Low Temperature[J]. J. Environ. Sci., 2021, 101: 1–15
Jiang L J, Liu Q C, Ran G J, et al. V2O5-Modified Mn-Ce/AC Catalyst with High SO2 Tolerance for Low-Temperature NH3-SCR of NO[J]. Chem. Eng. J., 2019, 370: 810–821
Kwon D W, Nam K B, Hong S C. The Role of Ceria on the Activity and SO2 Resistance of Catalysts for the Selective Catalytic Reduction of NOx by NH3[J]. Appl. Catal. B-Environ., 2015, 166: 37–44
Zhuang K, Zhang Y P, Huang T J, et al. Sulfur-Poisoning and Thermal Reduction Regeneration of Holmium-Modified Fe-Mn/TiO2 Catalyst for Low-Temperature SCR[J]. Journal of Fuel Chemistry and Technology, 2017, 45(11): 1 356–1 364
Liu F D, Shan W P, Lian Z H, et al. The Smart Surface Modification of Fe2O3 by WOx for Significantly Promoting the Selective Catalytic Reduction of NOx with NH3[J]. Appl. Catal. B-Environ., 2018, 230: 165–176
Yang W W, Liu F D, Xie L J, et al. Effect of V2O5 Additive on the SO2 Resistance of a Fe2O3/AC Catalyst for NH3-SCR of NOx at Low Temperatures[J]. Ind. Eng. Chem. Res., 2016, 55(10): 2 677–2 685
Yang S J, Liu C X, Chang H Z, et al. Improvement of the Activity of γ-Fe2O3 for the Selective Catalytic Reduction of NO with NH3 at High Temperatures: NO Reduction versus NH3 Oxidization[J]. Ind. Eng. Chem. Res., 2013, 52(16): 5 601–5 610
Xie C Y, Sun Y L, Zhu B Z, et al. Adsorption Mechanism of NH3, NO, and O2 Molecules over the FexOy/AC Catalyst Surface: a DFT-D3 Study[J]. New. J. Chem., 2021, 45(6): 3 169–3 180
Liang H, Zha X B, Gui K T, et al. A Study of Selective Catalysis Reduction Denitration Performance and Adsorption of NH3 and NO over γ-Fe2O3 Catalys[J]. Proceedings of the Csee, 2014, 34(32): 5 734–5 740
Shi X B, Chu B X, Wang F, et al. Mn-modified CuO, CuFe2O4 and γ-Fe2O3 Three-Phase Strong Synergistic Coexistence Catalyst System for NO Reduction by CO with Wider Active Window[J]. ACS Appl. Mater. Interfaces., 2018, 10(47): 40 509–40 522
Yao G H, Fang W, Wang X B, et al. Magnetic Field Effects on Selective Catalytic Reduction of NO by NH3 over Fe2O3 Catalyst in a Magnetically Fluidized Bed[J]. Energy, 2010, 35(5): 2 295–2 300
Xie S Y, Li C X, Wang G, et al. Enhancement of NO Catalytic Oxidation on Activated Carbon at Room Temperature by Nitric Acid Hydrothermal Treatment[J]. Appl. Surf. Sci., 2019, 471: 633–644
Wang D, Zhang X L, Peng J S, et al. Effects of Calcination Temperature on Selective Catalytic Reduction of NOx over γ-Fe2O3 Catalysts Prepared with Microwave Assistance[J]. Research of Environmental Sciences, 2015, 28(5): 808–815
Pasel J, Käßner P, Montanari B, et al. Transition Metal Oxides Supported on Active Carbons as Low Temperature Catalysts for the Selective Catalytic Reduction (SCR) of NO with NH3[J]. Appl. Catal. B-Environ., 1998, 18(3–4): 199–213
Zhan S H, Zhang H, Zhang Y, et al. Efficient NH3-SCR Removal of NOx with Highly Ordered Mesoporous WO3(χ)-CeO2 at Low Temperatures[J]. Appl. Catal. B-Environ., 2017, 203: 199–209
Dandekar A, Baker R T K, Vannice M A. Characterization of Activated Carbon, Graphitized Carbon Fibers and Synthetic Diamond Powder using TPD and Drifts[J]. Carbon, 1998, 36(12): 1 821–1 831
Palomo J, Ternero-Hidalgo J J, Rosas J M, et al. Selective Nitrogen Functionalization of Phosphorus-containing Activated Carbons[J]. Fuel Process. Technol., 2017, 156: 438–445
Sharma A, Dutta R K, Roychowdhury A, et al. Cobalt Doped CuO Nanoparticles as a Highly Efficient Heterogeneous Catalyst for Reduction of 4-Nitrophenol to 4-Aminophenol[J]. Appl. Catal. A-Gen., 2017, 543: 257–265
Yu L, Zhou H Q, Sun J Y, et al. Cu Nanowires Shelled with Ni-Fe Layered Double Hydroxide Nanosheets as Bifunctional Electrocatalysts for Overall Water Splitting[J]. Energ. Environ. Sci., 2017, 10(8): 1 820–1 827
Liu Z, Wang Z J, Qing S J, et al. Improving Methane Selectivity of Photo-induced CO2 Reduction on Carbon Dots Through Modification of Nitrogen-Containing Groups and Graphitization[J]. Appl. Catal. B-Environ., 2018, 232: 86–92
Yu J H, So J H. Synthesis and Characterization of Nitrogen-Containing Hydrothermal Carbon with Ordered Mesostructure[J]. Chem. Phys. Lett., 2019, 716: 237–246
Deng L L, Lu B Q, Li J L, et al. Effect of Pore Structure and Oxygen-Containing Groups on Adsorption of Dibenzothiophene over Activated Carbon[J]. Fuel, 2017, 200: 54–61
Lin Y T, Li Y R, Xu Z C, et al. Transformation of Functional Groups in the Reduction of NO with NH3 over Nitrogen-Enriched Activated Carbons[J]. Fuel, 2018, 223: 312–323
Yu Y X, Tan W, An D Q, et al. Insight into the SO2 Resistance Mechanism on γ-Fe2O3 Catalyst in NH3-SCR Reaction: A Collaborated Experimental and DFT Study ScienceDirect[J]. Appl. Catal. B-Environ., 2021, 281: 119 544
Wang L Y, Cheng X X, Wang Z Q, et al. Investigation on Fe-Co Binary Metal Oxides supported on Activated Semi-Coke for NO Reduction by CO[J]. Appl. Catal. B-Environ., 2017, 201: 636–651
Huang B F, Shi Z, Yang Z Y, et al. Mechanism of CO Selective Catalytic Reduction Denitration on Fe-Mn/AC Catalysts at Medium and Low Temperatures under Oxygen Atmosphere[J]. Chem. Eng. J., 2022, 446(4): 137 412
Pappas D K, Boningari T, Boolchand P, et al. Novel Manganese Oxide Confined Interweaved Titania Nanotubes for the Low-Temperature Selective Catalytic Reduction (SCR) of NOx by NH3[J]. J. Catal., 2016, 334: 1–13
Ge T T, Zhu B Z, Sun Y L, et al. Investigation of Low-Temperature Selective Catalytic Reduction of NOx with Ammonia over Cr-promoted Fe/AC Catalysts[J]. Environ. Sci. Pollut. R., 2019, 26(32): 33 067–33 075
Chen J Y, Zhu B Z, Sun Y L, et al. Investigation of Low-Temperature Selective Catalytic Reduction of NOx with Ammonia over Mn-Modified Fe2O3/AC Catalysts[J]. J. Braz. Chem. Soc., 2018, 29(1): 79–87
Yang J, Ren S, Zhang T S, et al. Iron Doped Effects on Active Sites Formation over Activated Carbon supported Mn-Ce Oxide Catalysts for Low-Temperature SCR of NO-ScienceDirect[J]. Chem. Eng. J., 2020, 379: 122 398
Zhu L, Zhang L, Qu H X, et al. A Study on Chemisorbed Oxygen and Reaction Process of Fe-CuOx/ZSM-5 via Ultrasonic Impregnation Method for Low-Temperature NH3-SCR[J]. J. Mol. Catal. A-Chem., 2015, 409: 207–215
Author information
Authors and Affiliations
Corresponding author
Additional information
Conflict of interest
All authors declare that there are no competing interests.
Funded by the General Project of Science and Technology Plan of Yunnan Science and Technology Department (Nos.202001AT070029, 2019FB077), Open Fund of Key Laboratory for Ferrous Metallurgy and Resources Utilization of Ministry of Education (No. FMRUlab-20-4)
Rights and permissions
About this article
Cite this article
Yang, Z., Huang, B., Zhang, G. et al. Low-temperature Denitration Mechanism of NH3-SCR over Fe/AC Catalyst. J. Wuhan Univ. Technol.-Mat. Sci. Edit. 38, 475–484 (2023). https://doi.org/10.1007/s11595-023-2721-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11595-023-2721-5