Abstract
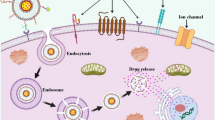
To design a new type of antitumor nanodrug carrier with good biocompatibility, a drug delivery system with a 2.19% drug-loading rate, measured by high-performance liquid chromatography (HPLC), was prepared by membrane hydration using a mixed polymer: Pluronic® F-127, which binds folic acid (FA), Pluronic® F-68 and triptolide (TPL) (FA-F-127/F-68-TPL). As a control, another drug delivery system based on a single polymer (FA-F-127-TPL) with a 1.90% drug-loading rate was prepared by substituting F-68 with F-127. The average particle sizes of FA-F-127/F-68-TPL and FA-F-127-TPL measured by a particle size analyzer were 30.7 nm and 31.6 nm, respectively. Their morphology was observed by atomic force microscopy (AFM). The results showed that FA-F-127-TPL self-assembled into nanomicelles, whereas FA-F-127/F-68-TPL self-assembled into nanogels. An MTT assay showed that a very low concentration of FA-F-127/F-68-TPL or FA-F-127-TPL could significantly inhibit the proliferation of multidrug-resistant (MDR) breast cancer cells (MCF-7/ADR cells) and induce cell death. The effects were signifcantly different from those of free TPL (P < 0.01). Using the fuorescent probe Nile red (Nr) as the drug model, FA-F-127/F-68-Nr nanogels and FA-F-127-Nr nanomicelles were prepared and then incubated with human hepatocarcinoma (HepG2) and MCF-7/ADR cells, and the fluorescence intensity in the cells was measured by a multifunctional microplate reader. The results indicated that both FA-F-127/F-68-Nr and FA-F-127-Nr had sustained release in the cells, but HepG2 and MCF-7/ADR cells exhibited significantly higher endocytosis of FA-F-127/F-68-Nr than that of FA-F-127-Nr (P < 0.01). A nude mice transplanted tumor model was prepared to monitor FA-F-127/F-68-Nr in the tumor tissue and organs by whole-body fluorescent imaging. The results showed that FA-F-127/F-68-Nr targeted tumor tissues. The prepared nanogels had small particle size, were easy to swallow, exhibited slow release property, targeted tumor cells, and could improve the antitumor effects of TPL; hence, they are ideal carriers for low-dose antineoplastic drugs.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Li Y, Maciel D, Rodrigues J, et al. Biodegradable Polymer Nanogels for Drug/Nucleic Acid Delivery[J]. Chemical Reviews, 2015, 115: 8564–8608
Sina N, Hugh R, Joselito M, et al. Progress Toward Robust Polymer Hydrogels[J]. Australian Journal of Chemistry, 2011, 64: 1007–1025
Song CK, Balakrishnan P, Shim CK et al. Enhanced in vitro Cellular Uptake of P-gp Substrate by Poloxamer-modifed Liposomes (PMLs) in MDR Cancer Cells[J] Journal of Microencapsulation, 2011, 28: 575–581
Hanke U, May K, Rozehnal V, et al. Commonly Used Nonionic Surfactants Interact Differently with the Human Effux Transporters ABCB1 (p-glycoprotein)and ABCC2 (MRP2)[J]. European Journal of Pharmaceutics and Biopharmaceutics, 2010, 76: 260–268
Li WN, Sun JL, Guan QX, et al. Research Progress of Pluronic as a Drug Delivery System for Reversing Multidrug Resistance of Tumors[J]. Chinese Pharmaceutical Journal, 2016, 51: 1270–1273
Mu L Q, Hu X B, Hu YY, et al. Advances in the Study of Overcoming Multidrug Resistance in Cancer by Pluronic Block Copolymers[J]. Chinese Journal of Hospital Pharmacy, 2014, 34: 1780–1784
Joung YK, Jang JY, Choi JH, et al. Heparin-Conjugated Pluronic Nanogels as Multi-Drug Nanocarriers for Combination Chemotherapy-[J]. Molecular Pharmaceutics, 2013, 10: 685–693
Kim JY, Choi WI, Kim M, et al. Tumor-targeting Nanogel That Can Function Independently for Both Photodynamic and Photothermal Therapy and Its Synergy from the Procedure of PDT Followed by PTT[J]. Journal of Controlled Release, 2013, 171: 113–121
Nguyen DH, Lee JS, Bae JW, et al. Targeted Doxorubicin Nanotherapy Strongly Suppressing Growth of Multidrug Resistant Tumor in Mice[J]. International Journal of Pharmaceutics, 2015, 495: 329–335
Merino S, Martín C, Kostarelos K, et al. Nanocomposite Hydrogels: 3D Polymer-Nanoparticle Synergies for On-Demand Drug Delivery[J]. ACS Nano, 2015, 9: 4686–4697
Ahmed E M. Hydrogel: Preparation, Characterization, and Applications: A Review[J]. Journal of Advanced Research, 2015, 6: 105–121
Sasaki Y, Akiyoshi K. Nanogel Engineering for New Nanobiomaterials: from Chaperoning Engineering to Biomedical Applications[J]. Chemical Record, 2010, 10: 366–376
Zhou QY, Zhang ZH, Pan JF, et al. Development of Poloxamers Used as Carriers for New Dosage Forms of Hydrophobic Drugs[J]. Chinese Journal of Modern Applied Pharmacy, 2011, 28: 315–319
Longmire M, Choyke PL, Kobayashi H. Clearance Properties of Na-no-sized Particles and Molecules as Imaging Agents: Considerations and Caveats[J]. Nanomedicine, 2008, 3: 703–717
Zhao P, Zheng M, Yue C, et al. Improving Drug Accumulation and Photothermal Efficacy in Tumor Depending on Size of ICG Loaded Lipid-Polymer Nanoparticles[J]. Biomaterials, 2014, 35: 6037–6046
Perrault SD, Walkey C, Jennings T, et al. Mediating Tumor Targeting Effciency of Nanoparticles Through Design[J]. Nano Letters, 2009, 9: 1909–1915
Lee H, Fonge H, Hoang B, et al. The Effects of Particle Size and Molecular Targeting on the Intratumoral and Subcellular Distribution of Polymeric Nanoparticles[J]. Molecular Pharmaceutics, 2010, 7: 1195–1208
Tao Y, Zhang ML, Ma PC, et al. Triptolide Inhibits Proliferation and Induces Apoptosis of Human Melanoma A375 Cells[J]. Asian Pacifc Journal of Cancer Prevention, 2012, 13: 1611–1615
Li H, Hui L, Xu W, et al. Modulation of P-glycoprotein Expression by Triptolide in Adriamycin-Resistant K562/A02 Cells[J]. Oncology Letters, 2012, 3: 485–489
Clawson K A, Borja-Cacho D, Antonoff M B, et al. Triptolide and TRAIL Combination Enhances Apoptosis in Cholangiocarcinoma[J]. Journal of Surgical Research, 2010, 163: 244–249
Johnson SM, Wang X, Evers BM. Triptolide Inhibits Proliferation and Migration of Colon Cancer Cells by Inhibition of Cell Cycle Regulators and Cytokine Receptors[J]. Journal of Surgical Research, 2011, 168: 197–205
Alakhova D Y, Rapoport N Y, Batrakova E V, et al. Differential Metabolic Responses to Pluronic in MDR and Non-MDR Cells: A Novel Pathway for Chemosensitization of Drug Resistant Cancers[J]. Journal of Controlled Release, 2010, 142: 89–100
Agarwal R, Jurney P, Raythatha M, et al. Effect of Shape, Size, and Aspect Ratio on Nanoparticle Penetration and Distribution inside Solid Tissues Using 3D Spheroid Models[J]. Advanced Healthcare Materials, 2015, 15: 2269–2280
Author information
Authors and Affiliations
Corresponding author
Additional information
Funded by the National Natural Science Foundation of Hubei Province (No. 2014CFB306), the National Natural Science Foundation of China(No. 51772233), the National Key Research and Development Program of China (No. 2016YFC1101605) and the Science and Technology Support Program of Hubei Province (No. 2015BAA085)
Rights and permissions
About this article
Cite this article
Yin, M., Su, Z., Cui, B. et al. A New Type of Nanogel Carrier based on Mixed Pluronic Loaded with Low-Dose Antitumor Drugs. J. Wuhan Univ. Technol.-Mat. Sci. Edit. 34, 960–967 (2019). https://doi.org/10.1007/s11595-019-2144-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11595-019-2144-5