Abstract
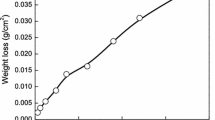
Corrosion behavior of 300M in neutral corrosion environments containing NaCl simulated by total immersion (TI), salt spraying (SS) and periodic immersion (PI), was investigated by surface analysis techniques, corrosion weight-loss method, and electrochemical measurements. In total immersion environment, rust on the steel consisted of a porous outer rust layer with main constituent of γ-FeOOH, and an inner rust layer of dense Fe3O4 film with network broad cracks. In salt spraying environment, outer rust with main composition of γ-FeOOH/α-FeOOH/Fe3O4 was compact, and inner rust showed dense Fe3O4 film. Rust formed by periodic immersion exhibited a compact outer rust layer with constituent of α-FeOOH/γ-FeOOH/Fe3O4 and an inner rust layer with composition of α-FeOOH/α-Fe2O3; inner rust showed a ultra-dense film adherent to the steel. The corrosion rate showed a rule of v ss(salt spraying)>v ti(total immersion)>>v pi(periodic immersion) in 0–240 h, and v ss≈v ti»v pi in 240–720 h. The rust formed by periodic immersion was dense and compact, with stable electrochemical properties, and had excellent protection on the steel. Humidity and oxygen concentration in all the environments played major roles in rust formation.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Solis M, William M. Defense Management Opportunities to Reduce Corrosion Costs and Increase Readiness: GAO-03-753[R]. USA, United States General Accounting Office, 2003
Liu YG, Li MQ, Luo J. The Modeling of Dynamic Recrystallization in the Isothermal Compression of 300M Steel[J]. Mater. Sci. Eng. A, 2013, 574(1): 1–8
Tomita Y, Okawa T. Effect of Microstructure on Mechanical Properties of Isothermally Bainite-transformed 300M Steel[J]. Mater. Sci. Eng. A, 1993, 172(1-2): 45–51
Figueroa D, Robinson MJ. The Effects of Sacrificial Coatings on Hydrogen Embrittlement and Re-embrittlement of Ultra High Strength Steels[J]. Corros. Sci., 2008, 50(4): 1066–1079
Zhang SS, Li MQ, Liu YG, et al. The Growth Behavior of Austenite Grain in the Heating Process of 300M Steel[J]. Mater. Sci. Eng. A, 2011, 528(15): 4967–4972
Luo J, Li MQ, Liu YG, et al. The Deformation Behavior in Isothermal Compression of 300M Ultrahigh-strength Steel[J]. Mater. Sci. Eng. A, 2012, 534: 314–322
García KE, Morales AL, Barrero SA, et al. New Contributions to the Understanding of Rust Layer Formation in Steels Exposed to a Total Immersion Test[J]. Corros. Sci., 2006, 48(9): 2813–2830
Pérez FR, Barrero CA, Hight Walker AR, et al. Effects of Chloride Concentration, Immersion Time, and Steel Composition on the Spinel Phase Formation[J]. Mater. Chem. Phys., 2009, 117(1): 214–223
Pérez FR, Barrero CA, García KE. Factors Affecting the Amount of Corroded Iron Converted into Adherent Rust in Steels Submitted to Immersion Tests[J]. Corro. Sci., 2010, 52(8): 2582–2591
Pérez FR, Barrero CA, Arnache O, et al. Structural Properties of Iron Phases Formed on Low Alloy Steels Immersed in Sodium Chloriderich Solutions[J]. Physica B: Condensed Matter, 2009, 404(8-11): 1347–1353
Sun M, Xiao K, Dong CF, et al. Electrochemical Behaviors of Ultra High Strength Steels with Corrosion Products[J]. Acta Metal Sin, 2011, 47(4): 442(in Chinese)
Sun M, Xiao K, Dong CF, et al. Electrochemical Corrosion Behavior of 300M Ultra High Strength Steel in Chloride Containing Environment[J]. Acta. Metall. Sin. (Engl. Lett. ), 2010, 23(4): 301–311
Yu M, Qi JY, Liu JH, et al. Corrosion Behaviors of Ultra-high Strength Steel 40CrNi2Si2MoVA in Submerged Zone of Simulated Seawater[J]. Corro. Pro., 2011, 32(10): 778 (in Chinese)
Hao XL, Liu JH, Li SM, et al. Effect of Neutral Salt Spray Precorrosion on Fatigue Life of AF1410 Steel[J]. J. Aeronaut. Mater., 2010, 30(1): 67–71(in Chinese)
Liu JH, Shang HB, Tao BW, et al. Corrosion Behavior of High Strength Steels 0Cr18Ni5 and AF1410[J]. Mater. Eng., 2004, 8: 29–31(in Chinese)
Wang ST, Yang SW, Gao KW, et al. Corrosion Behavior and Variation of Apparent Mechanical Property of a Novel Low Carbon Bainitic Steel in Environment Containing Chloride Ions[J]. Acta Metall. Sin., 2008, 44(9): 1116–1124(in Chinese)
Nishimura T, Katayama H, Noda K, et al. Effect of Co and Ni on the Corrosion Behavior of Low Alloy Steels in Wet/Dry Environments[J]. Corros. Sci., 2000, 42(9): 1611–1621
Liu JH, Tian S, Li SM, et al. Stress Corrosion Cracking of New Ultrahigh Strength Steel[J]. Acta Aeronaut Astronaut Sin., 2011, 32(6): 1164–1170(in Chinese)
Castaño JG, Botero CA, Restrepo AH, et al. Atmorpheric Corrosion of Carbon Steel in Colombia[J]. Corros. Sci., 2010(52): 216–223
Han SC. Atlas of Microstructure on Metals Corrosion[M]. Beijing: National Defense Industry Press, 2008: 11 (in Chinese)
Dubois F, Mendibide C, Paginier T, et al. Raman Mapping of Corrosion Products Formed onto Spring Steels During Salt Spray Experiments. A Correlation between the Scale Compostion and the Corrosion Resistance[J]. Corros. Sci., 2008, 50(12): 3401–3409
Zhang X, Xiao K, Dong, CF, et al. In situ Raman Spectroscopy Study of Corrosion Products on the Surface of Carbon Steel in Solution Containing Cl-and SO4 2-[J]. Eng. Fail. Anal., 2011,18: 1981–1989
Ma YT, Li Y, Wang FH. Corrosion of Low Carbon Steel in Atmospheric Environments of Different Chloride Content[J]. Corros. Sci., 2009, 51(5): 997–1006
Author information
Authors and Affiliations
Corresponding author
Additional information
Funded by the National Natural Science Foundation of China(No.51171011)
Rights and permissions
About this article
Cite this article
Guo, Q., Liu, J., Yu, M. et al. Corrosion behavior of ultra-high strength steel 300M in different simulated marine environments. J. Wuhan Univ. Technol.-Mat. Sci. Edit. 31, 372–378 (2016). https://doi.org/10.1007/s11595-016-1378-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11595-016-1378-8