Abstract
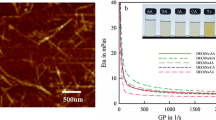
Light weight and mechanically strong α-chitin aerogels were fabricated using the sol-gel/self-assembly method from α-chitin in different aqueous alkali hydroxide (KOH, NaOH and LiOH)/urea solutions. All of the α-chitin solutions exhibited temperature-induced rapid gelation behavior. 13C nuclear magnetic resonance (NMR) spectra revealed that the aqueous alkali hydroxide/ urea solutions are non-derivatizing solvents for α-chitin. Fourier transform infrared (FT-IR), X-ray diffraction (XRD) and cross-polarization magic angle spinning (CP/MAS) 13C NMR confirmed that α-chitin has a stable aggregate structure after undergoing dissolution and regeneration. Subsequently, nanostructured α-chitin aerogels were fabricated by regeneration from the chitin solutions in ethanol and then freeze-drying from t-BuOH. These α-chitin aerogels exhibited high porosity (87% to 94%), low density (0.09 to 0.19 g/cm3), high specific surface area (419 to 535 m2/g) and large pore volume (2.7 to 3.8 cm3/g). Moreover, the α-chitin aerogels exhibited good mechanical properties under compression and tension models. In vitro studies showed that mBMSCs cultured on chitin hydrogels have good biocompatibility. These nanostructured α-chitin aerogels may be useful for various applications, such as catalyst supports, carbon aerogel precursors and biomedical materials.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Nair LS, Laurencin CT. Prog Polym Sci, 2007, 32: 762–798
Wang S, Lu A, Zhang LN. Prog Polym Sci, 2016, 53: 169–206
Bartlett DH, Azam F. Science, 2005, 310: 1775–1777
Wan ACA, Tai BCU. Biotechnol Adv, 2013, 31: 1776–1785
Arun KR, Sivashanmugam A, Deepthi S, Iseki S, Chennazhi KP, Nair SV, Jayakumar R. ACS Appl Mater Interf, 2015, 7: 9399–9409
Rinaudo M. Prog Polym Sci, 2006, 31: 603–632
Liu XX, Wang YF, Zhang NZ, Shanks RA, Liu HS, Tong Z, Chen L. Chin J Polym Sci, 2014, 32: 108–114
Yu YY, Guo L, Wang W, Wu J, Yuan Z. Sci China Chem, 2015, 58: 1866–1874
Pillai C, Paul W, Sharma CP. Prog Polym Sci, 2009, 34: 641–678
Yan N, Chen X. Nature, 2015, 524: 155–157
Ifuku S, Saimoto H. Nanoscale, 2012, 4: 3308–3318
Austin PR. Solvents for and purification of chitin. US Patent, 3892731. 1975-07-01
Sannan T, Kurita K, Iwakura Y. Die Makromolekulare Chemie, 1975, 176: 1191–1195
Feng F, Liu Y, Hu K. Carbohyd Res, 2004, 339: 2321–2324
Einbu A, Naess SN, Elgsaeter A, Vårum KM. Biomacromolecules, 2004, 5: 2048–2054
Tamura H, Nagahama H, Tokura S. Cellulose, 2006, 13: 357–364
Gagnaire D, Germain JS, Vincendon M. Die Makromolekulare Chemie, 1982, 183: 593–601
Kumar MNR. React Funct Polym, 2000, 46: 1–27
Xie H, Zhang S, Li S. Green Chem, 2006, 8: 630–633
Wu Y, Sasaki T, Irie S, Sakurai K. Polymer, 2008, 49: 2321–2327
Qin Y, Lu X, Sun N, Rogers RD. Green Chem, 2010, 12: 968–971
Sharma M, Mukesh C, Mondal D, Prasad K. RSC Adv, 2013, 3: 18149–18155
Mukesh C, Mondal D, Sharma M, Prasad K. Carbohyd Polym, 2014, 103: 466–471
Ladet S, David L, Domard A. Nature, 2008, 452: 76–79
Shi Z, Gao H, Feng J, Ding B, Cao X, Kuga S, Wang Y, Zhang L, Cai J. Angew Chem Int Ed, 2014, 53: 5380–5384
Torres JG, Femmer T, Laporte LD, Tigges T, Rahimi K, Gremse F, Zafarnia F, Lederle W, Ifuku S, Wessling M. Adv Mater, 2015, 27: 2989–2995
Rejinold SN, Chennazhi KP, Tamura H, Nair SV, Rangasamy J. ACS Appl Mater Interf, 2011, 3: 3654–3665
Shen X, Shamshina JL, Berton P, Gurau G, Rogers RD. Green Chem, 2016, 18: 53–75
Cai J, Zhang L. Macromol Biosci, 2005, 5: 539–548
Hu X, Du Y, Tang Y, Wang Q, Feng T, Yang J, Kennedy JF. Carbohyd Polym, 2007, 70: 451–458
Chang C, Chen S, Zhang L. J Mater Chem, 2011, 21: 3865–3871
Fan M, Hu Q. Carbohyd Res, 2009, 344: 944–947
Ding B, Cai J, Huang J, Zhang L, Chen Y, Shi X, Du Y, Kuga S. J Mater Chem, 2012, 22: 5801–5809
Duan B, Gao H, He M, Zhang L. ACS Appl Mater Interf, 2014, 6: 19933–19942
Huang Y, Zhong Z, Duan B, Zhang L, Yang Z, Wang Y, Ye Q. J Mater Chem B, 2014, 2: 3427–3432
Duan B, Zheng X, Xia Z, Fan X, Guo L, Liu J, Wang Y, Ye Q, Zhang L. Angew Chem Int Ed, 2015, 54: 1–6
Fang Y, Duan B, Lu A, Liu M, Liu H, Xu X, Zhang L. Biomacromolecules, 2015, 16: 1410–1417
Cai J, Huang J, Zhang L. Solvent compounds for chitin dissolution. China Patent, 201310034088.4. 2013-04-24
Terbojevich M, Carraro C, Cosani A, Marsano E. Carbohyd Res, 1988, 180: 73–86
Heux L, Brugnerotto J, Desbrières J, Versali MF, Rinaudo M. Biomacromolecules, 2000, 1: 746–751
Okuyama K, Noguchi K, Miyazawa T, Yui T, Ogawa K. Macromolecules, 1997, 30: 5849–5855
Nijenhuis KT, Winter HH. Macromolecules, 1989, 22: 411–414
Cai J, Zhang L. Biomacromolecules, 2006, 7: 183–189
Arvidson S, Lott J, Mc Allister J, Zhang J, Bates F, Lodge T, Sammler R, Li Y. Macromolecules, 2012, 46: 300–309
Minke R, Blackwell J. J Mol Biol, 1978, 120: 167–181
Focher B, Naggi A, Torri G, Cosani A, Terbojevich M. Carbohyd Polym, 1992, 17: 97–102
Sikorski P, Hori R, Wada M. Biomacromolecules, 2009, 10: 1100–1105
Ogawa Y, Kimura S, Wada M, Kuga S. J Struct Biol, 2010, 171: 111–116
Tanner SF, Chanzy H, Vincendon M, Roux JC, Gaill F. Macromolecules, 1990, 23: 3576–3583
Cárdenas G, Cabrera G, Taboada E, Miranda SP. J Appl Polym Sci, 2004, 93: 1876–1885
Kameda T, Miyazawa M, Ono H, Yoshida M. Macromol Biosci, 2005, 5: 103–106
Brugnerotto J, Lizardi J, Goycoolea F, Argüelles-Monal W, Desbrieres J, Rinaudo M. Polymer, 2001, 42: 3569–3580
Massiot D, Touzo B, Trumeau D, Coutures J, Virlet J, Florian P, Grandinetti P. Solid State Nucl Mag, 1996, 6: 73–83
Tsioptsias C, Michailof C, Stauropoulos G, Panayiotou C. Carbohydr Polym, 2009, 76: 535–540
Silva SS, Duarte ARC, Carvalho AP, Mano JF, Reis RL. Acta Biomaterialia, 2011, 7: 1166–1172
Heath L, Zhu L, Thielemans W. ChemSusChem, 2013, 6: 537–544
Tsutsumi Y, Koga H, Qi ZD, Saito T, Isogai A. Biomacromolecules, 2014, 15: 4314–4319
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
11426_2016_205_MOESM1_ESM.doc
Light weight, mechanically strong and biocompatible α-chitin aer-ogels from different aqueous alkali hydroxide/urea solutions
Rights and permissions
About this article
Cite this article
Ding, B., Zhao, D., Song, J. et al. Light weight, mechanically strong and biocompatible α-chitin aerogels from different aqueous alkali hydroxide/urea solutions. Sci. China Chem. 59, 1405–1414 (2016). https://doi.org/10.1007/s11426-016-0205-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-016-0205-5