Summary
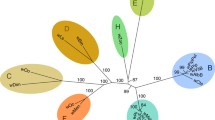
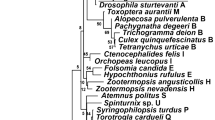
Wolbachia harbored by most filarial parasites, is critical to both embryogenesis and microfilarial development, and may lead to inflammation and pathogenesis in infected hosts. Based on alignment of the sequences from the wsp, ftsZ, and 16S rRNA genes, Wolbachia was demonstrated to exist in Angiostrongylus cantonensis, a non-filaroid nematode. Although the wsp gene may not be the best candidate for evolutionary analysis of Wolbachia, this gene has been sequenced from a broader coverage of the host species, making it feasible to be used for phylogenetic analysis in this study. The results from both Neighbor-joining and Maximum parsimony methods showed that this novel Wolbachia does not belong to any of the known groups (C or D) of nematode-derived Wolbachia. In addition, the wsp gene sequence of this newly identified endosymbiont revealed a high degree of identity (98%) with that from Diaea circumlita c2, tentatively classified into the putative group G. This suggests that Wolbachia from A. cantonensis could represent a deeply branched lineage in Wolbachia evolution or the occurrence of horizontal transfer between infected hosts. In conclusion, the findings provide some insights into our understanding of the evolution of Wolbachia, particularly the isolate from A. cantonensis.
Similar content being viewed by others
References
O’Neill S.L., Pettigrew M.M., Sinkins S.P., Braig H.R., Andreadis T.G., Tesh R.B. (1997) In vitro cultivation of Wolbachia pipientis in an Aedes albopictus cell line. Insect Mol. Biol. 6:33–39
Werren J.H. (1997) Biology of Wolbachia. Annu. Rev. Entomol. 42:587–609
O’Neill S.L., Giordano R., Colbert A.M.E., Karr T.L., Robertson H.M. (1992) 16S rRNA phylogenetic analysis of the bacterial endosymbionts associated with cytoplasmic incompatibility in insects. Proc. Nat. Acad. Sci. USA 89:2699–2702
Bazzocchi C., Jamnongluk W., O’Neill S.L., Anderson T.J.C., Genchi C., Bandi C. (2000) wsp gene sequences from the Wolbachia of filarial nematodes. Curr. Microbiol. 41:96–100
Anderson C.L., and Karr T.L., Wolbachia: evolutionary novelty in a rickettsial bacteria. BMC Evo. Biol. 1:http://www.biomedcentral.com/1471-2148/1/10, 2001
Sironi M., Bandi C., Sacchi L., Di Sacco B., Damiani G., Genchi C. (1995) Molecular evidence for a close relative of the arthropod endosymbiont Wolbachia in a filarial worm. Mol. Biochem. Parasitol. 74:223–227
Taylor M.J., Bilo K., Cross H.F., Archer J.P. and Underwood A.P., (1999) 16S rDNA phylogeny and ultrastructural characterization of Wolbachia intracellular bacteria of the filarial nematodes Brugia malayi, B. pahangi, and Wuchereria bancrofti. Exp. Parasitol. 91:356–361
Büttner D.W., Wanji S., Bazzocchi C., Bain O., and Fischer␣P., Obligatory symbiotic Wolbachia endobacteria are absent from Loa loa. Filaria J. 2:http://www.filariajournal.com/content/2/1/10, 2003
McGarry H.F., Pfarr K., Egerton G., Hoerauf A., Akue J., Enyong P., Wanji S., Klager S.L., Bianco A.E., Beeching N.J. and Taylor M.J., Evidence against Wolbachia symbiosis in Loa loa. Filaria J. 2:http://www.filariajournal.com/content/2/1/9, 2003
Hise A.G., Gillette-Ferguson I., Pearlman E. (2004) The role of endosymbiotic Wolbachia bacteria in filarial disease. Cell. Microbiol. 6:97–104
Peixoto C.A., Silva L.F., Teixeira K.M., Rocha A. (2001) Ultrastructural characterization of intracellular bacteria of Wuchereria bancrofti. Trans. R. Soc. Trop. Med. Hyg. 95:566–568
Brattig N.W. (2004) Pathogenesis and host responses in human onchocerciasis: impact of Onchocerca filariae and Wolbachia endobacteria. Microbes Infect. 6:113–128
McGarry H.F., Egerton G.L., Taylor M.J. (2004) Population dynamics of Wolbachia bacterial endosymbionts in Brugia malayi. Mol. Biochem. Parasitol. 135:57–67
Taylor M.J., Bandi C., Hoerauf A., Lazdins J. (2000) Wolbachia bacteria of filarial nematodes: a target for control? Parasitol. Today 16:179–180
Brattig N.W., Büttner D.W., Hoerauf A. (2001) Neutrophil accumulation around Onchocerca worms and chemotaxis of neutrophils are dependent on Wolbachia endobacteria. Microbes Infect. 3:439–446
André A.v.S., Blackwell N.M., Hall L.R., Hoerauf A., Brattig N.W., Volkmann L., Taylor M.J., Ford L., Hise A.G., Lass J.H., Diaconu E., Pearlman E. (2002) The role of endosymbiotic Wolbachia bacteria in the pathogenesis of river blindness. Science 295:1892–1895
Zhou W., Rousset F., O’Neill S.L. (1998) Phylogeny and PCR-based classification of Wolbachia strains using wsp gene sequences. Proc. R. Soc. Lon. Ser. B 265:509–515
Tsai H.C., Liu Y.C., Kunin C.M., Lee S.S., Chen Y.S., Lin H.H., Tsai T.H., Lin W.R., Huang C.K., Yen M.Y., Yen C.M. (2001) Eosinophilic meningitis caused by Angiostrongylus cantonensis: report of 17 cases. Am. J. Med. 111:109–114
Wang L.C., Wan Y.L. (2004) Alteration of antibodies against the fifth-stage larvae and changes in brain magnetic resonance images in experimentally infected rabbits with Angiostrongylus cantonensis. J. Parasitol. 90:1193–1196
Casiraghi M., Favia G., Cancrini G., Bartoloni A., Bandi C. (2001) Molecular identification of Wolbachia from the filarial nematode Mansonella ozzardi. Parasitol. Res. 87:417–420
Tsai K.H., Lien J.C., Huang C.G., Wu W.J., Chen W.J. (2004) Molecular (sub-)grouping of the endosymbiont Wolbachia infection among mosquitoes of Taiwan. J. Med. Entomol. 41:677–683
Thompson J.D., Higgins D.G., Gibson T.J. (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position specific gap penalties and weight matrix choice. Nucleic Acids Res. 22:4673–4680
Kumar S., Tamura K., Jakobsen I.B., Nei M. (2001) MEGA2: Molecular evolutionary genetics analysis software. Arizona State University, Tempe, AZ, USA
Lo N., Casiraghi M, Salati E, Bazzocchi C, Bandi C. (2002) How many Wolbachia supergroups exist? Mol. Biol. Evol. 19:341–346
Tsai K.H., Phylogeny of the symbiont Wolbachia in mosquitoes and its potential interactions with arbovirus in the mosquito. PhD dissertation, National Taiwan University, Taipei, Taiwan, 2004, pp.117
Taylor M.J., Hoerauf A. (1999) Wolbachia bacteria of filarial nematodes. Parasitol. Today 5:437–442
Bordenstein S.R., Fitch D.H., Werren J.H. (2003) Absence of Wolbachia in nonfilariid nematodes. J. Nematol. 35:266–270
Blaxter M.L., de Ley P., Garey J.R., Liu L.X., Scheldeman P., Vierstraete A., Vanfleteren J.R., Mackey L.Y., Dorris M., Frisse L.M., Vida J.T., Thomas W.K. (1998) A molecular evolutionary framework for the phylum Nematoda. Nature 392:71–75
Bandi C., Anderson T.J.C., Genchi C., Blaxter M.L. (1998) Phylogeny of Wolbachia in filarial nematodes. Proc. R. Soc. Lond. Ser. B 265:2407–2413
Casiraghi M., Bain O., Guerrero R., Martin C., Pocacqua V., Gardner S.L., Franceschi A., Bandi C. (2004) Mapping the presence of Wolbachia pipientis on the phylogeny of filarial nematodes: evidence for symbiont loss during evolution. Intl. J. Parasitol. 34:191–203
Hong X.Y., Gotoh T., Noda H. (2002) Sensitivity comparison of PCR primers for detecting Wolbachia in spider mites. Appl. Entomol. Zool. 37:379–383
Egyed Z., Sréter T., Széll Z., Nyirő G., Dobos-Kovács M., Márialigeti K., Varga I. (2004) Electron microscopic and molecular identification of Wolbachia endosymbionts from Onchocerca lupi: implications for therapy. Vet. Parasitol. 106:75–82
Werren J.H., Bartos J.D. (2001) Recombination in Wolbachia. Curr. Biol. 11:431–435
Werren J.H., Zhang W., Guo L.R. (1995) Evolution and phylogeny of Wolbachia: reproductive parasites of arthropods. Proc. R. Soc. Lon. Ser. B 261:55–63
Casiraghi M., Anderson T.J., Bandi C., Bazzocchi C., Genchi C. (2001) A phylogenetic analysis of filarial nematodes: comparison with the phylogeny of Wolbachia endosymbionts. Parasitology 122:93–103
Vandekerckhove T.T.M., Watteyne S., Willems A., Swings J.G., Mertens J., Gillis M. (1999) Phylogenetic analysis of the 16S rDNA of the cytoplasmic bacterium Wolbachia from the novel host Folsomia candida (Hexapoda, Collembola) and its implications for wolbachial taxonomy. FEMS Microbiol. Lett. 180:279–286
Bandi C., Trees A.J., Brattig N.W. (2001) Wolbachia in filarial nematodes: evolutionary aspects and implications for the pathogenesis and treatment of filarial diseases. Vet. Parasitol. 98:215–238
Rowley S.M., Raven R.J., McGraw E.A. (2004) Wolbachia pipiens in Australian spiders. Curr. Microbiol. 49:208–214
Punkosdy G.A., Dennis V.A., Lasater B.L., Tzertzinis G., Foster J.M., Lammie P.J. (2001) Detection of serum IgG antibodies specific for Wolbachia surface protein in rhesus monkeys infected with Brugia malayi. J. Infect. Dis. 184:385–389
Kanpittaya J., Jitpimolmard S., Tiamka S., Mairiang E. (2000) MR findings of eosinophilic meningoencephalitis attributed to Angiostrongylus cantonensis. Am. J. Neuroradiol. 21:1090–1094
Pien F.D., Pien B.C. (1999) Angiostrongylus cantonensis eosinophilic meningitis. Intl. J. Infect. Dis. 3:161–163
Aoki M., Sugaya H., Ishida K., Yoshimura K. (1998) The role of CD4+ and CD8+ T-cells in host morbidity and innate resistance to Angiostrongylus cantonensis in the mouse. Parasitol. Res. 84:91–99
Taylor M.J. (2000) Wolbachia bacteria of filarial nematodes in the pathogenesis of disease and as a target for control. Trans. R. Soc. Trop. Med. Hyg. 94:596–598
Alto W. (2001) Human infections with Angiostrongylus cantonensis. Pac. Hlth. Dialog. 8:176–182
Mohanty B.P. (2001) Wolbachia: potential targets for control of filariases. Curr. Sci. 80:614–616
Acknowledgments
This work was financially supported by a grant from the National Science Council of Taiwan (NSC95-2320-B-182-017) and partially supported by Chang Gung Memorial Hospital (CMRP 150251).
Author information
Authors and Affiliations
Corresponding author
Additional information
Kun-Hsien Tsai and Chin-Gi Huang are contributed equally to this study
Rights and permissions
About this article
Cite this article
Tsai, KH., Huang, CG., Wang, LC. et al. Molecular evidence for the endosymbiont Wolbachia in a non-filaroid nematode, Angiostrongylus cantonensis . J Biomed Sci 14, 607–615 (2007). https://doi.org/10.1007/s11373-007-9181-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11373-007-9181-3