Summary
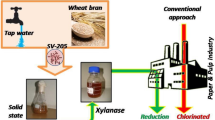
The production of xylanase from Bacillus coagulans has been studied with respect to the environmental parameters, the carbon source and the concentration of carbon source at the shake flask level. Among the various carbon sources used, wheat straw powder favoured higher enzyme production. Xylan isolated from wheat straw gave higher enzyme production as compared to the birchwood xylan. Maximum enzyme activity of 165 IU/ml was obtained with 2% wheat straw xylan in a shake flask study. Improvement of xylanase production was achieved by increasing the wheat straw powder concentration up to 3%. Enzyme has optimum activity at a temperature of 55 °C and pH of 7. The concentrated crude enzyme was found to reduce the kappa number of enzyme-treated eucalyptus pulp by␣5.45% with a marginal increase in the CED viscosity of the enzyme treated pulp as compared to the non-enzymatically treated pulp.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Avcioglu B., Eyupoglu B., Baker U., 2005 Production and characterization of xylanase of a Bacillus strain isolated from soil World Journal of Microbiology and Biotechnology 21: 65–68
Bailey M.J., Biely M., Poutanen K., 1992 Labaratory testing methods for assay of xylanase activity Journal of Biotechnology 23: 257–270
Biely P., Mackenzie C.R., Schneider H., 1988 Methods in Enzymology 160A, Acedemic Press Inc. New York 700–707
Bocchini, D.A., Oliveira, O.M.M.F., Gomes, E. & Da Silva, R. 2005 Use of sugar cane bagasse and grass hydrolysates as carbon sources for xylanase production by Bacillus circulans D1 in submerged fermentation. Process Biochemistry 40, 3653–3659
Chauhan, S., Choudhury, B., Singh, S.N. & P. Ghosh 2005 Application of xylanase enzyme of Bacillus coagulans as a prebleaching agent on non-woody pulps. Process Biochemistry doi: 10.1016/j.procbio.2005.06.003
Damaso M.C., Almedia M.S., Kurtenbach E., Martins O.B., Pereira N. Jr., Andrade C.M., 2003 Optimized expression of a thermostable xylanase from Thermomyces lanuginosus in Pichia pastoris Applied and Environmental Microbiology 69: 6064–6072
Damino V.B., Bocchini D.A., Gomes E, Silva R.D., 2003 Application of crude xylanase from Bacillus licheniformis 77–2 to the bleaching of eucalyptus Kraft pulp World Journal of Microbiology and Biotechnology 19: 139–144
Dhillon A., Gupta J.K., Khanna S., 2000 Enhanced production, purification and characterization of a novel cellulase-poor thermo-stable alkalitolerant xylanase from Bacillus circulans AB16 Process Biochemistry 35: 849–856
Ding C.H., Jiang Z.Q., Li X.T., Li L.T., Kusakabe I., 2004 High activity of xylanase production by Streptomyces olivaceovirdis E-86 World Journal of Microbiology and Biotechnology. 20: 7–10
Gomes J., Gomes I., Walter S., 2000 Thermolabile xylanase of the Antarctic yeast Cryptococcus adeeliae: Production and properties Extremophiles 4: 227–235
Gorgenes, J.F., Van Zye, W.H., Kuoetze, J.H. & Han-Hageral, B. 2005 Amino acid supplementation improves heterologous protein production by Saccharomyces cerevisiae in defined medium. Applied Microbiology and Biotechnology 67, 684–691
Gupta S., Kuhad R.C., Bhushan B, Hoondal G.S., 2001 Improved xylanase production from a haloalkalophilic Staphylococcus sp Sg-13 using inexpensive agricultural residues World Journal of Microbiology and Biotechnology 17: 5–8
Heck J.X., Fl^ores S.H., Hertz P.F., Ayub MAZ., 2005 Optimization of cellulase-free xylanase activity produced by Bacillus coagulans BL69 in solid-state cultivation Process Biochemistry 40: 107–112
IUPAC (International Union of Pure, Applied Chemistry) 1987 measurement of cellulose activities. Pure and Applied Chemistry 59: 257–268
Miller G.L., 1959 Use of dinitro-salicylic acid reagent for determination of reducing sugars Analytical Chemistry 31: 426–428
Oliveria L.A., Barros N.B., Porto A.L., Tambourgi E.B., 2004 Xylanase by Penicillum ganthinellum in a poly(ethylene glycol/cashew nut tree gum) aqueous two-phase systems Biotechnology Progress 20: 1880–1884
Pandey P., Pandey A.K., 2002 Production of cellulose free thermostable xylanases by an isolated strain of Aspergillus niger PPI, utilizing various lignocellulosic wastes World Journal of Microbiology and Biotechnology 18: 281–283
Sá-Pereira P., Mesquita A., Duarte J.C., Barros M.R.A., Costa-Ferreira M, 2002 Rapid production of thermostable cellulase-free xylanase by a strain of Bacillus subtilis and its properties Enzyme and Microbial Technology 30: 924–933
Senthilkumar S.R., Ashokkumar B., Chandra R.K., Gunasekaran P., 2005 Optimization of medium composition for alkali-stable xylanase production by Aspergillus fischeri Fxn-1 in solid-state fermentation using central composite rotary design Bioresource Technology 96: 1380–1386
Subramaniyan S., Prema P., 2002 Biotechnology of microbial xylanases: Enzymology, molecular biology and application Critical Reviews in Biotechnology 22: 33–46
Subramaniyan S., Sandhia G.S., Prema P., 2001 Control of xylanase production without protease activity in Bacillus sp. by selection of nitrogen source Biotechnology Letters 23: 369–371
Techapun C., Poosaran N., Watanabe M., Sasaki K., 2003 Thermostable and alkaline tolerant microbial cellulase-free xylanase produced from agricultural wastes and the properties required for use in pulp bleaching bioprocesses: a review Process Biochemistry 38: 1327–1340
Acknowledgements
The work was financially supported by Indian paper Manufacturers Association, New Delhi. We are thankful to Central pulp and paper research Institute, Saharanpur, India for evaluating our enzyme on Kraft pulp.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Choudhury, B., Chauhan, S., Singh, S. et al. Production of Xylanase of Bacillus coagulans and its bleaching potential. World J Microbiol Biotechnol 22, 283–288 (2006). https://doi.org/10.1007/s11274-005-9033-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11274-005-9033-0