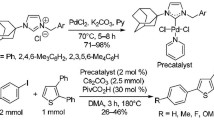
Abstract
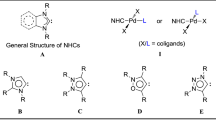
The palladium complexes of the Pd-PEPPSI type with N-heterocyclic carbenes of the 1,2,4-triazole series were synthesized in 76—99% yields by the reactions of PdCl2 with 1,4-di- alkyl-1,2,4-triazolium salts in pyridine in the presence of KBr or KI as sources of halide ions and tetrabutylammonium salts as phase-transfer catalysts. The obtained complexes can be used as efficient catalysts for the Suzuki—Miyaura cross-coupling and are not inferior to the commercially available Pd-PEPPSI catalysts in activity.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
W. A. Herrmann, M. Elison, J. Fischer, C. Köcher, G. R. J. Artus, Angew. Chem., Int. Ed., 1995, 34, 2371.
M. N. Hopkinson, C. Richter, M. Schedler, F. Glorius, Nature, 2014, 510,485.
P. G. Gildner, T. J. Colacot, Organometallics, 2015, 34, 5497.
C. I. Ezugwu, N. A. Kabir, M. Yusubov, F. Verpoort, Coord. Chem. Rev., 2016, 307, Part 2,188.
A. Nasr, A. Winkler, M. Tamm, Coord. Chem. Rev., 2016, 316,68.
N-Heterocyclic Carbenes: From Laboratory Curiosities to Efficient Synthetic Tools (2), The Royal Society of Chemistry, 2017, Doi: 10.1039/9781782626817-FP021, p. P021.
V. A. Glushkov, M. S. Valieva, O. A. Maiorova, E. V. Baigacheva, A. A. Gorbunov, Russ. J. Org. Chem., 2011, 47,230.
V. A. Glushkov, E. N. Teplykh, M. S. Denisov, A. A. Gorbunov, Russ. J. Org. Chem., 2012, 48,815.
V. N. Mikhaylov, V. N. Sorokoumov, I. A. Balova, Russ. Chem. Rev., 2017, 86,459.
B. V. Romanovsky, I. G. Tarkhanova, Russ. Chem. Rev., 2017, 86,444.
N. M. Shcheglova, V. D. Kolesnik, R. V. Ashirov, E. A. Krasnokutskaya, Russ. Chem. Bull., 2016, 65,490.
I. A. Utepova, A. A. Musikhina, O. N. Chupakhin, Russ. Chem. Bull., 2016, 65, 2523.
C. J. O’Brien, E. A. B. Kantchev, C. Valente, N. Hadei, G. A. Chass, A. Lough, A. C. Hopkinson, M. G. Organ, Chem. Eur. J., 2006, 12, 4743.
C. Valente, S. Çalimsiz, K. H. Hoi, D. Mallik, M. Sayah, M. G. Organ, Angew. Chem., Int. Ed., 2012, 51, 3314.
C. Valente, M. Pompeo, M. Sayah, M. G. Organ, Org. Process Res. Dev., 2014, 18,180.
R. D. J. Froese, C. Lombardi, M. Pompeo, R. P. Rucker, M. G. Organ, Acc. Chem. Res., 2017, 50,224.
C. Dash, M. M. Shaikh, P. Ghosh, Eur. J. Inorg. Chem., 2009, 2009, 1608.
A. Zanardi, J. A. Mata, E. Peris, Organometallics, 2009, 28, 4335.
A. Zanardi, J. A. Mata, E. Peris, J. Am. Chem. Soc., 2009, 131, 14531.
A. Kumar, M. K. Gangwar, A. P. Prakasham, D. Mhatre, A. C. Kalita, P. Ghosh, Inorg. Chem., 2016, 55, 2882.
A. V. Astakhov, O. V. Khazipov, A. Y. Chernenko, D. V. Pasyukov, A. S. Kashin, E. G. Gordeev, V. N. Khrustalev, V. M. Chernyshev, V. P. Ananikov, Organometallics, 2017, 36, 1981.
A. N. Kashin, I. P. Beletskaya, Russ. J. Org. Chem., 2011, 47,475.
A. N. Kashin, D. A. Panchuk, L. M. Yarysheva, A. L. Volynskii, I. P. Beletskaya, Russ. J. Org. Chem., 2011, 47,48.
N. A. Bumagin, V. I. Potkin, Russ. Chem. Bull., 2016, 65,321.
O. N. Gorunova, Y. K. Grishin, M. M. Ilyin, K. A. Kochetkov, A. V. Churakov, L. G. Kuz’mina, V. V. Dunina, Russ. Chem. Bull., 2017, 66,282.
T. G. Ermakova, N. P. Kuznetsova, A. S. Pozdnyakov, L. I. Larina, S. A. Korzhova, I. V. Mazyar, V. S. Shcherbakova, A. V. Ivanov, A. I. Mikhaleva, G. F. Prozorova, Russ. Chem. Bull., 2016, 65,485.
A. V. Astakhov, O. V. Khazipov, E. S. Degtyareva, V. N. Khrustalev, V. M. Chernyshev, V. P. Ananikov, Organometallics, 2015, 34, 5759.
S. Yaçar, Ç. ahin, M. Arslan, I. Özdemir, J. Organomet. Chem., 2015, 776,107.
D. Rottschäfer, C. J. Schürmann, J.-H. Lamm, A. N. Paesch, B. Neumann, R. S. Ghadwal, Organometallics, 2016, 35, 3421.
R. Maity, M. van der Meer, B. Sarkar, Dalton Trans., 2015, 44,46.
L. Benhamou, C. Besnard, E. P. Kündig, Organometallics, 2014, 33,260.
G. Wang, G. Liu, Y. Du, W. Li, S. Yin, S. Wang, Y. Shi, C. Cao, Trans. Met. Chem., 2014, 39,691.
H. Baier, A. Kelling, H. J. Holdt, Eur. J. Inorg. Chem., 2015, 2015, 1950.
R. Maity, A. Verma, M. van der Meer, S. Hohloch, B. Sarkar, Eur. J. Inorg. Chem., 2016, 2016,111.
E. Steeples, A. Kelling, U. Schilde, D. Esposito, New J. Chem., 2016, 40, 4922.
D. B. Eremin, V. P. Ananikov, Coord. Chem. Rev., 2017, 346,2.
T. G. G. Battye, L. Kontogiannis, O. Johnson, H. R. Powell, A. G. W. Leslie, Acta Crystallogr., Sect. D: Biol. Crystallogr., 2011, 67,271.
P. Evans, Acta Crystallogr., Sect. D: Biol. Crystallogr., 2006, 62,72.
G. Sheldrick, Acta Crystallogr., Sect. A: Found. Adv., 2015, 71, 3.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 1, pp. 0079—0084, January, 2018
Rights and permissions
About this article
Cite this article
Chernenko, A.Y., Astakhov, A.V., Pasyukov, D.V. et al. Pd-PEPPSI complexes based on 1,2,4-triazol-3-ylidene ligands as efficient catalysts in the Suzuki—Miyaura reaction. Russ Chem Bull 67, 79–84 (2018). https://doi.org/10.1007/s11172-018-2040-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-018-2040-8