Abstract
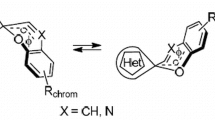
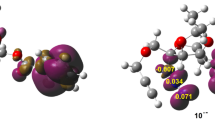
Structural and spectral characteristics of spiro[1,3,4]oxadiazines were calculated by density functional theory method (B3LYP/6-311++G(d,p)). It is shown that a fundamental difference of spirooxadiazines from known spirocyclic compounds is that the ring-opened form is more energetically favorable (by 4.9 kcal mol–1) compared to the ring-closed one. The closeness between the total energies of closed and open forms of spirooxadiazines suggests all isomers to coexist in solution. The intramolecular rearrangements of compounds under study associated with bond cleavage—formation are accompanied by overcoming of lower-energy barriers than those in the case of analogous spiropyrans and spiroxazines. The calculated electronic spectra (TD DFT) and overlapping absorption band maxima of the closed and open spirooxadiazine forms suggest a low probability of photoinitiated ring opening—closure in this compound.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Molecular Switches, Eds B. L. Feringa, W. R. Browne, Vol. 1—2, Wiley-VCH, Weinheim, Germany, 2011.
V. I. Minkin, Russ. Chem. Bull. (Int. Ed.), 2008, 57, 687 [Izv. Akad. Nauk, Ser. Khim., 2008, 673].
Techniques of Chemistry, Vol. III: Photochromism, Ed. G. H. Brown, Wiley-Interscience, New York, 1971, 853 pp.
Photochromism. Molecules and Systems, Eds H. Dürr, H. Bouas-Laurent, Elsevier, Amsterdam, 1990, 1044 pp.
Organic Photochromic and Thermochromic Compounds, Eds J. C. Crano, R. J. Guglielmetti, Vol. 1—2, Plenum Press, New York, 2000.
G. Bercovic, V. Krongauz, V. Weiss, Chem. Rev., 2000, 100, 1741.
V. Lokshin, A. Samat, A. V. Metelitsa, Russ. Chem. Rev., 2002, 71, 893.
V. I. Minkin, Chem. Rev., 2004, 104, 2751.
V. I. Minkin, Russ. Chem. Rev., 2013, 82, 1.
R. Klajn, Chem. Soc. Rev., 2014, 43, 148.
M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, J. A. Montgomery, T. Vreven, K. N. Kudin, J. C. Burant, J. M. Millam, S. S. Iyengar, J. Tomasi, V. Barone, B. Mennucci, M. Cossi, G. Scalmani, N. Rega, G. A. Petersson, H. Nakatsuji, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, M. Klene, X. Li, J. E. Knox, H. P. Hratchian, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, P. Y. Ayala, K. Morokuma, G. A. Voth, P. Salvador, J. J. Dannenberg, V. G. Zakrzewski, S. Dapprich, A. D. Daniels, M. C. Strain, O. Farkas, D. K. Malick, A. D. Rabuck, K. Raghavachari, J. B. Foresman, J. V. Ortiz, Q. Cui, A. G. Baboul, S. Clifford, J. Cioslowski, B. B. Stefanov, G. Liu, A. Liashenko, P. Piskorz, I. Komaromi, R. L. Martin, D. J. Fox, T. Keith, M. A. Al-Laham, C. Y. Peng, A. Nanayakkara, M. Challacombe, P. M. W. Gill, B. Johnson, W. Chen, M. W. Wong, C. Gonzalez, J. A. Pople, GAUSSIAN 03, Revision E.01, Gaussian, Inc., Wallingford (CT), 2004.
W. Kohn, L. J. Sham, Phys. Rev., 1965, 140, A1133.
A. D. J. Becke, J. Chem. Phys., 1993, 98, 5648.
V. I. Minkin, A. G. Starikov, R. M. Minyaev, A. A. Starikova, Theor. Exp. Chem. (Engl. Transl.), 2011, 46, 363 [Teor. Eksperim. Khim., 2010, 46, 352].
A. A. Starikova, R. M. Minyaev, A. G. Starikov, V. I. Minkin, Dokl. Chem. (Engl. Transl.), 2013, 453, 263 [Dokl. Akad. Nauk Khim., 2013, 453, 401].
A. A. Starikova, R. M. Minyaev, A. G. Starikov, V. I. Minkin, Eur. J. Inorg. Chem., 2013, 4203.
R. M. Minyaev, Russ. Chem. Rev., 1994, 63, 883.
V. Barone, M. Cossi, J. Phys. Chem. A, 1998, 102, 1995.
Chemcraft, version 1.7, 2013: http://www.chemcraftprog.com.
S. Nakamura, K. Uchida, A. Murakami, M. Irie, J. Org. Chem., 1993, 58, 5543.
T. Horii, Y. Abe, R. Nakao, J. Photochem. Photobiol. A, 2001, 144, 119.
V. I. Minkin, A. V. Metelitsa, I. V. Dorogan, B. S. Lukyanov, S. O. Besugliy, J.-C. Micheau, J. Phys. Chem. A, 2005, 109, 9605.
F. Maurel, J. Aubard, M. Rajzmann, R. Guglielmetti, A. Samat, J. Chem. Soc., Perkin Trans., 2002, 2 1307.
A. Perrier, F. Maurel, E. A. Perpte, V. Wathelet, D. Jacquemin, J. Phys. Chem. A, 2009, 113, 13004.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to the Academician of the Russian Academy of Sciences N. S. Zefirov on the occasion of his 80th birthday.
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 1, pp. 0040—0046, January, 2016.
Rights and permissions
About this article
Cite this article
Minkin, V.I., Starikova, A.A. Computer simulation of the isomerization mechanism and spectral characteristics of spiro[1,3,4]oxadiazines. Russ Chem Bull 65, 40–46 (2016). https://doi.org/10.1007/s11172-016-1262-x
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-016-1262-x