Abstract
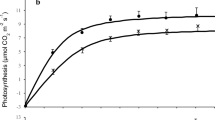
In crowns of chestnut trees the absorption of radiant energy is not homogeneous; leaves from the south (S) side are the most irradiated, but leaves from the east (E) and west (W) sides receive around 70 % and those from north (N) face less than 20 % of the S irradiation. Compared to the S leaves, those from the N side were 10 % smaller, their stomata density was 14 % smaller, and their laminae were 21 % thinner. N leaves had 0.63 g(Chl) m−2, corresponding to 93 % of total chlorophyll (Chl) amount in leaves of S side. The ratios of Chl a/b were 2.9 and 3.1 and of Chl/carotenoids (Car) 5.2 and 4.8, respectively, in N and S leaves. Net photosynthetic rate (P N) was 3.9 µmol(CO2) m−2 s−1 in S leaves, in the E, W, and N leaves 81, 77, and 38 % of that value, respectively. Morning time (10:00 h) was the period of highest P N in the whole crown, followed by 13:00 h (85 % of S) and 16:00 h with 59 %. Below 500 µmol m−2 s−1 of photosynthetic photon flux density (PPFD), N leaves produced the highest P N, while at higher PPFD, the S leaves were most active. In addition, the fruits from S side were 10 % larger than those from the N side.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- A:
-
leaf area
- ACMA:
-
9-amino-6-chloro-methoxyacridine
- ALM:
-
area leaf mass
- Car:
-
carotenoids
- Chl:
-
chlorophyll
- DM:
-
dry mass
- E:
-
transpiration rate
- g s :
-
stomatal conductance
- MV:
-
methylviologen
- P N :
-
net photosynthetic rate
- PPFD:
-
photosynthetic photon flux density
- Q:
-
ACMA fluorescence
- RWC:
-
relative water content
- SLA:
-
specific leaf area
- T L :
-
leaf temperature
- WUE:
-
water use efficiency
- Ψp :
-
leaf pressure potential
- Ψw :
-
leaf water potential
- Ψπ :
-
leaf osmotic potential
References
Almeida, P., Dinis, L.-T., Coutinho, J., Pinto, T., Anjos, R., Ferreira-Cardoso, J., Pimentel-Pereira, M., Peixoto, F., Gomes-Laranjo, J.: Effect of temperature and radiation on photosynthesis productivity in chestnut populations (Castanea sativa Mill. cv. Judia).-Acta agron. hung. 55: 193–203, 2007.
Anderson, J.M., Chow, W.S., Goodchild, D.J.: Thylakoid membrane organisation in sun/shade acclimation.-In: Evans, J.R., Caemmerer, S. von, Adams, W.W., III. (ed.): Ecology of Photosynthesis in Sun and Shade. Pp. 11–26. CSIRO, Melbourne 1988.
Boardman, N.K.: Comparative photosynthesis of sun and shade plants.-Annu. Rev. Plant Physiol. 28: 355–377, 1977.
Bounous, G.: Il castagno. [Chestnut.]-Edagricole, Bologna 2002. [In Ital.]
Dai, Z., Edwards, G.E., Ku, M.S.B.: Control of photosynthesis and stomatal conductance in Ricinus communis L. (castor bean) by leaf to air vapor pressure deficit.-Plant Physiol. 99: 1426–1434, 1992.
Fernández-López, J., Zas, R., Diaz, R., Aravanopoulos, F.A., Alizoti, P.G., Botta, R., Mellano, M.G., Villani, Cherbini, F. M., Eriksson, G.: Geographic variability among extreme European wild chestnut populations.-Acta Hort. 693: 181–186, 2005.
Ferreira-Cardoso, J.V.: [Characterization and Alternative Uses of the Fruits of Chestnut Tree (Castanea sativa Mill.): Chemical Composition, Nutritive Value and the Effect on Finishing Diets for Duroc Pigs.]-PhD Thesis. UTAD, Vila Real 2002. [In Port.]
Gomes-Laranjo, J., Coutinho, J.P., Ferreira-Cardoso, J., Pimentel-Pereira, M., Ramos, C., Torres-Pereira, J.: Assessment to a new concept of chestnut orchard management in vegetative wall.-Acta Hort. 693: 707–712, 2005.
Gomes-Laranjo, J.C.E.: [Photosynthesis in Chestnut: Integrated Study at Different Biological Organization Levels.]-PhD Thesis. UTAD, Vila Real 2001. [In Port.]
Gomes-Laranjo, J.C.E., Peixoto, F., Wong Fong Sang, H.W., Torres-Pereira, J.M.G.: Study of the temperature effect in three chestnut (Castanea sativa Mill.) cultivars’ behaviour.-J. Plant Physiol. 163: 945–955, 2006.
Gomes-Laranjo, J.C.E., Salgado, P., Wong Fong Sang, H.W., Kraayenhof, R., Torres-Pereira, J.M.G.: Isolation of chestnut chloroplasts: Membrane potentials of chestnut and spinach thylakoids.-Photosynthetica 43: 237–246, 2005.
Heiniger, U., Conedera, M.: Chestnut forests and chestnut cultivation in Switzerland.-Proceedings of the International Chestnut Conference. Pp. 175–178. West Virginia University, Morgantown 1992.
Kraayenhof, R., Sterk, G.J., Sang, H.W.W.F.: Probing biomembrane interfacial potential and pH profiles with a new type of float-like fluorophores positioned at varying distance from the membrane surface.-Biochemistry 32: 10057–10066, 1993.
Kraayenhof, R., Sterk, C.J., Sang, H.W.W.F., Krab, K., Epand, R.M.: Monovalent cations differentially affect membrane surface properties and membrane curvature, as revealed by fluorescent probes and dynamic light scattering.-Biochim. biophys. Acta 1282: 293–302, 1996.
Kubiske, M.E., Pregitzer, K.S.: Ecophysiological responses to stimulated canopy gaps of two tree species of contrasting shade tolerance in elevated CO2.-Funct. Ecol. 11: 24–32, 1997.
Kvet, J., Marshall, J.K.: Assessment of leaf area and other assimilating plant surfaces.-In: Šesták, Z., Čatský, J., Jarvis, P.G. (ed.): Plant Photosynthetic Production. Manual of Methods. Pp. 517–547. Dr W. Junk Publ., The Hague 1971.
Lawlor, D.W.: Photosynthesis: Molecular, Physiological and Environmental Processes. 2nd Ed.-Longman Scientific & Technical, Hong Kong 1993.
Lichtenthaler, H.K.: Differences in morphology and chemical composition of leaves grown at different light intensities and qualities.-In: Baker, N.R., Davies, W.J., Ong, C.K. (ed.): Control of Leaf Growth. Pp. 201–221. Cambridge University Press, Cambridge-London-New York-New Rochelle-Melbvourne-Sydney 1985.
Lichtenthaler, H.K.: Chlorophylls and carotenoids-pigments of photosynthetic biomembranes.-In: Colowick, S.P., Kaplan, N.O. (ed.): Methods in Enzymology. Vol. 148. Pp. 350–382. Academic Press, San Diego-New York-Berkeley-Boston-London-Sydney-Tokyo-Toronto 1987.
Lichtenthaler, H.K., Ač, A., Marek, M., Kalina, J., Urban, O.: Differences in pigment composition, photosynthetic rates and chlorophyll fluorescence images of sun and shade leaves of four tree species.-Plant Physiol. Biochem. 2007. doi:10.1016/j.plaphy epub ahead of print
Lichtenthaler, H.K., Babani, F.: Light adaptation and senescence of the photosynthetic apparatus. Changes in pigment composition, chlorophyll fluorescence parameters and photosynthetic activity.-In: Papageorgiou, G.C., Govindjee (ed.): Chlorophyll Fluorescence: A Signature of Photosynthesis. Pp. 713–736. Springer, Dordrecht 2004.
Lichtenthaler, H.K., Buschmann, C., Döll, M., Fietz, H.-J., Bach, T., Kozel, U., Meier, D., Rahmsdorf, U.: Photosynthetic activity, chloroplast ultrastructure, and leaf characteristics of high-light and low-light plants and of sun and shade leaves.-Photosynth. Res. 2: 115–141, 1981.
Lichtenthaler, H.K., Meier, D., Buschmann, C.: Development of chloroplasts at high and low light quanta fluence rates.-Isr. J. Bot. 33: 185–194, 1984.
Mills, H.A., Jones, J.B., Jr.: Plant Analysis Handbook II.-MicroMacro Publishing, Athens 1996.
Osmond, C.B., Chow, W.S.: Ecology of photosynthesis in the sun and shade: summary and prognostications.-In: Evans, J.R., Caemmerer, S. von, Adams, W.W., III (ed.): Ecology of Photosynthesis in Sun and Shade. Pp. 1–9. CSIRO, Melbourne 1988.
Packer, L., Torres-Pereira, J.M.G., Chang, P., Hansen, S.: Stabilization of chloroplast membranes as measured by light induced quenching of acridine dyes.-In: Avron, M. (ed.): Proceedings of the Third International Congress on Photosynthesis. Vol. II. Pp. 867–872. Elsevier, Amsterdam-Oxford-New York 1975.
Pearcy, R.W.: Acclimation to sun and shade.-In: Raghavendra, A.S. (ed.): Photosynthesis. A Comprehensive Treatise. Pp. 250–263. Cambridge University Press, Cambridge 1998.
Porra, R.J., Thompson, W.A., Kriedemann, P.E.: Determination of accurate extinction coefficients and simultaneous equations for assaying chlorophylls a and b extracted with four different solvents: verification of the concentration of chlorophyll standards by atomic absorption spectroscopy.-Biochim. biophys. Acta 975: 384–394, 1989.
Rottenberg, H.: The measurement of membrane potential and ΔpH in cells, organelles, and vesicles.-In: Packer, L., Fleischer, S. (ed.): Biomembranes. Pp. 625–648. Academic Press, San Diego 1997.
Salisbury, F.B., Ross, C.W.: Plant Physiology.-Wadsworth Publishing Company, Belmont 1992.
Scholander, P., Hammel, H.T., Bradstreet, E.D., Hemmingsen, E.A.: Sap pressure in vascular plants: negative hydrostatic pressure can be measured in plants.-Science 148: 339–346, 1965.
Schuldiner, S., Rottenberg, H., Avron, M.: Determination of ΔpH in chloroplasts: 2. Fluorescence amines as a probe for the determination of ΔpH in chloroplasts.-Eur. J. Biochem. 25: 64–70, 1972.
Taiz, L., Zeiger, E.: Plant Physiology. 3rd Ed.-Sinauer Associates Publ., Sunderland 2002.
Thompson, W.A., Huang, L.-K., Kriedemann, P.E.: Photosynthetic response to light and nutrients in sun-tolerant and shade-tolerant rainforest trees. II. Leaf gas exchange and component processes of photosynthesis.-Aust. J. Plant Physiol. 19: 19–42, 1992a.
Thompson, W.A., Kriedemann, P.E., Craig, L.E.: Photosynthetic response to light and nutrients in sun-tolerant and shade-tolerant rainforest trees. I. Growth, leaf anatomy and nutrient content.-Aust. J. Plant Physiol. 19: 1–18, 1992b.
Torres-Pereira, J.M.G., Wong Fong Sang, H., Theuvenet, A.P.R., Kraayenhof, R.: Electric surface charge dynamics of chloroplast thylakoid membranes. Temperature dependence of electrokinetic potential and aminoacridine interaction.-Biochim. biophys. Acta 767: 295–303, 1984.
Turner, N.C.: Measurement of plant water status by the pressure chamber technique.-Irrig. Sci. 9: 289–308, 1988.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gomes-Laranjo, J., Coutinho, J.P., Galhano, V. et al. Differences in photosynthetic apparatus of leaves from different sides of the chestnut canopy. Photosynthetica 46, 63–72 (2008). https://doi.org/10.1007/s11099-008-0012-1
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11099-008-0012-1