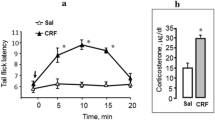
Corticotrophin-releasing factor (CRF) is involved in regulating pain sensitivity and can elicit analgesic effects in animals and humans. The aim of the present work was to investigate the involvement of types 1 and 2 CRF receptors (CRF-1 and CRF-2 receptors) in mediating the analgesic action of CRF on somatic pain sensitivity when given systemically to conscious rats. Somatic pain sensitivity was tested in terms of the latent period (LP) of the tailflick reaction in response to thermal stimulation (the tail flick test). The involvement of CRF-1 and CFR-2 receptors was studied by systemic administration of their specific antagonists NBI 27914 and astressin 2B, respectively. Systemic administration of CRF increased the latent period of the pain reaction (it had an analgesic effect). Prior administration of NBI 27914 or astressin 2B eliminated the analgesic effect of CRF. In addition, administration of NBI 27914 affected the basal latent period of the pain reaction, increasing it. These data provide evidence that the analgesic effect of CRF may be mediated by both CRF-1 and CRF-2 receptors. CRF-1 receptors, unlike CRF-2 receptors, may also be involved in regulating the basal level of pain sensitivity.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
N. I. Yarushkina and T. R. Bagaeva, “Mechanisms of the analgesic effect of corticotropin-releasing factor in conscious rats,” Ros. Fiziol. Zh., 96, No. 2, 128–137 (2010).
N. I. Yarushkina, T. R. Bagaeva, and L. P. Filaretova, “The analgesic effect of corticotropin-releasing factor (CRF) on somatic pain sensitivity: the involvement of glucocorticoids and CRF-2 receptors,” Ros. Fiziol. Zh., 94, No. 10, 1118–1125 (2008).
N. I. Yarushkina, T. R. Bagaeva, and L. P. Filaretova,, “Mechanisms of the analgesic action of corticotropin-releasing factor on pain sensitivity in rats,” Ros. Fiziol. Zh., 99, No. 7, 806–819 (2013).
R. E. Abdelhamid, K. J. Kovacs, J. D. Pasley, et al., “Forced swim-induced musculoskeletal hyperalgesia is mediated by CRF2 receptors but not by TRPV 1 receptors,” Neuropharmacology, 72, 29–37 (2013).
F. J. Ayesta and K. E. Nikolarakis, “Peripheral but not intracerebroventricular corticotropin-releasing hormone (CRH) produces antinociception which is not opioid mediated,” Brain Res., 503, No. 2, 219–224 (1989).
M. S. Cepeda, I. Bonney, J. Moyano, and D. B. Carr, “Corticotropin releasing hormone (CRH) produces analgesia in a thermal injury model independent of its effect on systemic beta-endorphin and corticosterone,” Regul. Pept., 118, No. 1–2, 39–43 (2004).
M. Dolatshahi-Somehsofla, S. Esmaeili-Mahani, F. Motamedi, et al., “Adrenalectomy potentiates the antinociceptive effects of calcium channel blockers,” Pharm. Biochem. Behav., 92, No. 2, 327–334 (2009).
Y. Fu and V. Neugebauer, “Differential mechanisms of CRF1 and CRF2 receptor functions in the amygdala in pain-related synaptic facilitation and behavior,” J. Neurosci., 28, No. 15, 3861–3876 (2008).
K. M. Hargreaves, C. D. Flores, and G. Mueller, “The role of pituitary β-endorphin in mediating CRF-induced antinociception,” Am. J. Physiol., 258, No. 1, E235–E242 (1990).
R. L. Hauger, V. Risbrough, O. Brauns, and F. M. Dautzenberg, “Corticotropin releasing factor (CRF) receptor signaling in the central nervous system: new molecular targets,” CNS Neurol. Disord. Drug Targets, 5, No. 4, 453–479 (2006).
M. Hummel, T. Cummons, P. Lu, et al., “Pain is salient ‘stressor’ that is mediated by corticotrophin-releasing factor-1 receptors,” Neurophar macology, 59, 160–166 (2010).
G. Ji and V. Negebauer, “Pro- and antinociceptive effects of corticotropin-releasing factor (CRF) in central amygdala neurons are mediated through different receptors,” J. Neurophysiol., 99, No. 3, 1201–1212 (2008).
G. Ji, Y. Fu, H. Adwanikar, and V. Negebauer, “Non-pain-related CRF1 activation in the amygdala facilitates synaptic transmission and pain responses,” Mol. Pain, 9, 2. DOI: 10.1186/1744-8069-9-2 (2013).
G. Ji, Y. Fu, K. A. Ruppert, and V. Negebauer, “Pain-related anxiety-like behavior requires CRF1 receptors in the amygdala,” Mol. Pain, 3, 13, DOI: 10.1186/1744-8069-433-13 (2013).
W. R. Lariviere and R. Melzack, “The role of corticotropin-releasing factor in pain and analgesia,” Pain, 84, No. 1, 1–12 (2000).
W. R. Lariviere, P. Fiorenzani, I. Ceccarelli, et al., “Central CRH administration changes formalin pain responses in male and female rats,” Brain Res., 1383, 128–134 (2011).
D Le Bars, M. Gozariu, and S. W. Cadden, “Animal models of nociception,” Pharmacol. Rev., 53, No. 4, 597–652 (2001).
R. Likar, S. A. Mousa, H. Steinkellner, et al., “Involvement of intra-articular corticotropin-releasing hormone in postoperative pain modulation,” Clin. J. Pain, 23, No. 2, 136–142 (2007).
V. Martinez, L. Wang, J. Rivier, et al., “Central CRF, urocortins and stress increase colonic transit via CRF1 receptors while activation of CRF2 receptors delays gastric transit in mice,” J. Physiol., 556, No. 1, 221–234 (2004).
R. Matejec, H. Uhlick, C. Hotz, et al., “Corticotropin-releasing hormone reduces pressure pain sensitivity in humans without involvement of beta-endorphin (1-31), but does not reduce heat pain sensitivity,” Neuroendocrinology, 82, No. 3–4, 185–197 (2005).
T. Miguel and R. Nunes-de-Souza, “Anxiogenic and antinociceptive effects of corticotrophin-releasing factor (CRF) injections into periaqueductal gray are modulated by CRF1 receptor in mice,” Horm. Behav., 60, No. 3, 292–300 (2011).
S. A. Mousa, C. P. Bopaiah, C. Stein, and M. Schafer, “Involvement of corticotropin-releasing hormone receptor subtypes 1 and 2 in peripheral opioid-mediated inhibition of inflammatory pain,” Pain, 106, No. 3, 297–307 (2003).
S. A. Mousa, C. P. Bopaiah, J. F. Richter, et al., “Inhibition of inflammatory pain by CRF at peripheral, spinal and supraspinal sites: involvement of areas coexpressing CRF receptors and opioid peptides,” Neuropsychopharmacology, 32, No. 12, 2530–2542 (2007).
M. Nijsen, N. Ongenae, A. Meulemans, and B. Coulie, “Divergent role for CRF 1 and CRF2 receptors in the modulation of visceral pain,” Neurogastroenterol. Motil., 17, No. 3, 423–432 (2005).
T. Nozu and M. Kudaira, “Corticotropin-releasing factor induces rectal hypersensitivity after repetitive painful rectal distention in healthy humans,” J. Gastroenterol., 41, 740–744 (2006).
C. L. Rivier, D. E. Grigoriadis, and J. E. Rivier, “Role of corticotropin-releasing factor receptors type 1 and 2 in modulating the rat adrenocorticotropin response to stressors,” Endocrinology, 144, No. 6, 2396–2403 (2003).
D. Roosterman, T. George, S. W. Schneider, et al., “Neuronal control of skin function: the skin as a neuroimmunoendocrine organ,” Physiol. Rev., 86, No. 4, 1309–1379 (2006).
T. Rouwette, P. Vanelderen, E. W. Roubos, et al., “The amygdala, a relay station for switching on and off pain,” Eur. J. Pain, 16, 782–792 (2012).
S. Samuelsson, J. S. Lange, R. T. Hinkle, et al., “Corticotropin-releasing factor 2 receptor localization in skeletal muscle,” J. Histochem. Cytochem, 52, No. 7, 967–977 (2004).
Y. Tache, “Stress activates corticotropin releasing factor signaling pathways,” in: Chemistry and Molecular Aspects of Drug Design and Action, E. A. Rekka and P. N. Kouronakis (eds.), CRC Press (2008), pp. 75–90.
J. P. Vit, D. J. Clauw, T. Moallem, et al., “Analgesia and hyperalgesia from CRF receptor modulation in the central nervous system of Fischer and Lewis rats,” Pain, 121, No. 3, 241–260 (2006).
N. I. Yarushkina, A. I. Bogdanov, and L. P. Filaretova, “Somatic pain sensitivity during formation and healing of acetic acid-induced gastric ulcers in conscious rats,” Auton. Neurosci. Basic Clin., 126-127, No. 1–2, 100–105 (2006).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Rossiiskii Fiziologicheskii Zhurnal imeni I. M. Sechenova, Vol. 100, No. 11, pp. 1287–1296, November, 2014.
Rights and permissions
About this article
Cite this article
Yarushkina, N.I., Bagaeva, T.R. & Filaretova, L.P. Effects of Corticotropin-Releasing Factor (CRF) on Somatic Pain Sensitivity in Conscious Rats: Involvement of Types 1 and 2 CRF Receptors. Neurosci Behav Physi 46, 472–477 (2016). https://doi.org/10.1007/s11055-016-0260-7
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11055-016-0260-7