Abstract
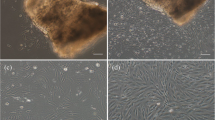
Three cell lines derived from fin (WSF), head soft tissue (WSHST) and body muscle (WSBM) were established from white sturgeon (Acipenser transmontanus). Characterization included determination of optimal growth kinetics, karyotyping, and mitochondrial ribosomal RNA (rRNA) genotyping. The primary cultures of these cells were generated by the explant technique using the L-15 medium supplemented with 20% fetal bovine serum and epidermal/fibroblast growth factors. The cells grew between 15–30 °C, but optimal growth occurred at 25 °C with a doubling time of 48 hours. The cell lines can be readily maintained in-vitro and have been subcultured over 35 times. Following cryopreservation in liquid nitrogen, thawed cells exhibit a viability of > 90% after a 16-month storage period. Chromosomal typing of these cell lines at their 17th passage revealed a chromosomal distribution of 242 to 278 with an apparent peak ranging from 250 to 260. Polymerase chain reaction amplification of mitochondrial 16S ribosomal RNA and sequence analysis indicated 100% identity of the sequences found in the cell lines with those found in the source animal, confirming that the cell lines were of A. transmontanus origin.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Birstein VJ, Doukakis P, DeSalle R (2002). Molecular phylogeny of Acipenseridae: non-monophyly of scaphirhynchinae. Copeia 2:287–301.
Bronzi P, Rosenthal H, Arlati G, Williot P (1999). A brief overview on the status and prospects of sturgeon farming in Western and Central Europe. J Appl Ichthyol 15:224–227.
Earley EM (1975). Chromosome preparations from monolayer cell cultures. TCA Manual 1:31–35
Georgiadis MP, Hedrick RP, Carpenter TE, Gardner IA (2001). Factors influencing transmission, onset and severity of outbreaks due to white sturgeon iridovirus in a commercial hatchery. Aquaculture 194:21–35.
Fantana F (1994). Chromosomal nucleolar orga-nizing regions in four sturgeon species as markers of karyotype evolution in Acipenseriformes (Pisces). Genome,37:888–892.
Fantana F, Lanfredi M, Rossi R, Bronzi P, Arlati G (1995). Established cell lines from three sturgeon species. Sturg Quart 3(4):6–7.
Freshney RI ed (1994). Culture of Animal Cells, A Manual of Basic Techniques. New York: 486 pp.
Hashiguchi K, Ikushima T (1998). Nucleotide changes in mitochondrial 16S rRNA gene from different mammalian cell lines. Genes Genet Syst 75: 317–321.
Hedrick RP, Speas J, Kent ML, McDowell T (1985). Adeno-like virus associated with a disease of cultured white sturgeon (Acipenser transmonotanus). Canadian J Fish Aquat Sci 42:1321–1325.
Hedrick RP, Groff JM, McDowell TS, Wingfield WH (1990). An iridovirus from the integument of white sturgeon. Dis Aquat Organ 8:39–44.
Hedrick RP, McDowell TS, Groff JM, Yun S, Wingfield WH (1991a). Isolation of an epithelio-tropic herpesvirus from white sturgeon Acipenser transmontanus. Dis Aquat Organ 11:49–56.
Hedrick RP, McDowell T, RosemarkR, Aronstein D, Lannan CN (1991b). Two cell lines from white sturgeon. Trans Am Fish Soc 120:528–534.
Krieger J, Fuerst PA (2002). Evidence of multiple alleles of the nuclear 18S ribosomal RNA gene in sturgeon (Family:Acipenseridae). J Appl Ichthyol 18:290–297.
Kshirsagar SG, Patole MS, Shouche YS (1997). Characterization of insect cell lines:heteroduplex analysis employing a mitochondrial 16S ribosomal RNA gene fragment. Anal Biochem 253:65–69.
LaPatra SE, Groff JM, Jones GR, Munn B, Patterson TL, Holt RA, Hauck AK, Hedrick RP (1994). Occurrence of white sturgeon iridovirus infections among cultured white sturgeon in the Pacific Northwest. Aquaculture 126:201–210.
LaPatra SE, Ireland S, Groff JM, Clemens K, Siple J (1999)Adaptive disease management strategies for the endangered population of Kootenai River white sturgeon Acipenser transmontanus. Fisheries 24:6–14.
Li MF, Marrayatt V, Annand C, Odense P (1985). Fish cell culture:two newly developed cell lines from Atlantic sturgeon (Acipenser oxyrhynchus) and gup-py (Poecilia reticulata). Can J Zool 63:2867–2874.
Lu Y, Aguirre AA, Hamm C, Wang Y, Yu Q, Loh PC, Yanagihara R (2000). Establishment, cryopreserva-tion, and growth of 11 cell lines prepared from ajuvenile Hawaiian monk seal, Monachus schauins-landi. Meth cell sci. 22:115–124.
Sudeep AB, Mourya DT, Shouche YS, Pidiyar V, Pant U (2002a). A new cell line from the embryonic tissue of helicoverpa armigera HBN (Lepidoptera:Noctuidae). In Vitro cell Dev Biol Anim 38:262–264.
Sudeep AB, Shouche YS, Mourya DT, Pant U (2002b). New helicoverpa armigera Hbn cell line from larval hemocyte for baculovirus studies. Indian J Exp Biol 40:69–73.
Van Eenennaam AL, Murray JD, Medrano JF (1998a). Synaptonemal complex analysis in spermatocytes of white sturgeon, Acipenser transmontanus Richardson (Pisces,Acipenseridae), a fish with a very high chromosome number. Genome 41:51–61.
Van Eenennaam AL, Murray JD, Medrano JF (1998b). Mitotic analysis of the North American white sturgeon, Acipenser transmontanus Richardson (Pisces, Acipenseridae), a fish with a very high chromosome number. Genome. 41:266–271.
Watson LR, Yun S, Groff JM, Hendrick RP (1955). Characteristics and pathogenicity of a novel herpesvirus isolated from adult and subadult white sturgeon Acipenser transmontanus. Dis Aquat Organ 22:199–210.
Watson LR, GroFF JM, Hedrick RP (1998). Replication and pathogenesis of white sturgeon iridovirus (WSIV)in experimentally infected white sturgeon Acipenser transmontanus juveniles and sturgeon cell lines. Dis Aquat Organ 32:173–184.
Williot P, Sabeau L, Gessner J, Arlati G, Bronzi P, Gulyas T et al.(2001). Sturgeon farming in western Europe:recent developments and perspectives. Aquat. Living Resour 14:367–374.
Wolf K (1988). Fish viruses and viral diseases. Ithaca, New York:Cornell University Press.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, G., Lapatra, S., Zeng, L. et al. Establishment, growth, cryopreservation and species of origin identification of three cell lines from white sturgeon, Acipenser transmontanus. Methods Cell Sci 25, 211–220 (2004). https://doi.org/10.1007/s11022-004-9120-x
Revised:
Issue Date:
DOI: https://doi.org/10.1007/s11022-004-9120-x