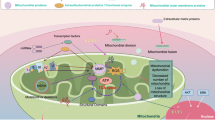
Abstract
Mitochondria have an essential role in powering cells by generating ATP following the metabolism of pyruvate derived from glycolysis. They are also the major source of generating reactive oxygen species (ROS), which have regulatory roles in cell death and proliferation. Mutations in mitochondrial DNA (mtDNA) and dysregulation of mitochondrial metabolism have been frequently described in human tumors. Although the role of oxidative stress as the consequence of mtDNA mutations and/or altered mitochondrial functions has been demonstrated in carciongenesis, a causative role of mitochondria in tumor progression has only been demonstrated recently. Specifically, the subject of this mini-review focuses on the role of mitochondria in promoting cancer metastasis. Cancer relapse and the subsequent spreading of cancer cells to distal sites are leading causes of morbidity and mortality in cancer patients. Despite its clinical importance, the underlying mechanisms of metastasis remain to be elucidated. Recently, it was demonstrated that mitochondrial oxidative stress could actively promote tumor progression and increase the metastatic potential of cancer cells. The purpose of this mini-review is to summarize current investigations of the roles of mitochondria in cancer metastasis. Future development of diagnostic and therapeutic strategies for patients with advanced cancer will benefit from the new knowledge of mitochondrial metabolism in epithelial cancer cells and the tumor stroma.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Brandon, M., Baldi, P. and Wallace, D.C. (2006). Mitochondrial mutations in cancer. Oncogene 25, 4647-62
Chen, E.I. et al. (2007). Adaptation of energy metabolism in breast cancer brain metastases. Cancer Res 67, 1472-86
Chen, X., Li, J., Hou, J., Xie, Z. and Yang, F. (2010). Mammalian mitochondrial proteomics: insights into mitochondrial functions and mitochondria-related diseases. Expert Rev Proteomics 7, 333-45
Desouki, M.M., Kulawiec, M., Bansal, S., Das, G.M. and Singh, K.K. (2005). Cross talk between mitochondria and superoxide generating NADPH oxidase in breast and ovarian tumors. Cancer Biol Ther 4, 1367-73
Edlund, M., Sung, S.Y. and Chung, L.W. (2004). Modulation of prostate cancer growth in bone microenvironments. J Cell Biochem 91, 686-705.
Eng, C., Kiuru, M., Fernandez, M.J. and Aaltonen, L.A. (2003). A role for mitochondrial enzymes in inherited neoplasia and beyond. Nat Rev Cancer 3, 193-202
Espineda, C.E., Chang, J.H., Twiss, J., Rajasekaran, S.A. and Rajasekaran, A.K. (2004). Repression of Na,K-ATPase beta1-subunit by the transcription factor snail in carcinoma. Mol Biol Cell 15, 1364-73
Falkenberg, M., Larsson, N.G. and Gustafsson, C.M. (2007). DNA replication and transcription in mammalian mitochondria. Annu Rev Biochem 76, 679-99
Feld, R., Rubinstein, L.V. and Weisenberger, T.H. (1984). Sites of recurrence in resected stage I non-small-cell lung cancer: a guide for future studies. J Clin Oncol 2, 1352-8
Fidler, I.J. (2003). The pathogenesis of cancer metastasis: the 'seed and soil' hypothesis revisited. Nat Rev Cancer 3, 453-8
Goh, J., Enns, L., Fatemie, S., Hopkins, H., Morton, J., Pettan-Brewer, C. and Ladiges, W. (2011). Mitochondrial targeted catalase suppresses invasive breast cancer in mice. BMC Cancer 11, 191
Gupta, G.P. and Massague, J. (2006). Cancer metastasis: building a framework. Cell 127, 679-95
Hess, K.R., Varadhachary, G.R., Taylor, S.H., Wei, W., Raber, M.N., Lenzi, R. and Abbruzzese, J.L. (2006). Metastatic patterns in adenocarcinoma. Cancer 106, 1624-33
Hoffman, P.C., Mauer, A.M. and Vokes, E.E. (2000). Lung cancer. Lancet 355, 479-85
Ishikawa, K. et al. (2008). ROS-generating mitochondrial DNA mutations can regulate tumor cell metastasis. Science 320, 661-4
Isidoro, A., Martinez, M., Fernandez, P.L., Ortega, A.D., Santamaria, G., Chamorro, M., Reed, J.C. and Cuezva, J.M. (2004). Alteration of the bioenergetic phenotype of mitochondria is a hallmark of breast, gastric, lung and oesophageal cancer. Biochem J 378, 17-20
Karbowski, M. (2010). Mitochondria on guard: role of mitochondrial fusion and fission in the regulation of apoptosis. Adv Exp Med Biol 687, 131-42
Karrison, T.G., Ferguson, D.J. and Meier, P. (1999). Dormancy of mammary carcinoma after mastectomy. J Natl Cancer Inst 91, 80-5
Kulawiec, M., Owens, K.M. and Singh, K.K. (2009). Cancer cell mitochondria confer apoptosis resistance and promote metastasis. Cancer Biol Ther 8, 1378-85
Lambert, A.J. and Brand, M.D. (2009). Reactive oxygen species production by mitochondria. Methods Mol Biol 554, 165-81
Lee, H.C. et al. (2005). Mitochondrial genome instability and mtDNA depletion in human cancers. Ann N Y Acad Sci 1042, 109-22
Mambo, E., Chatterjee, A., Xing, M., Tallini, G., Haugen, B.R., Yeung, S.C., Sukumar, S. and Sidransky, D. (2005). Tumor-specific changes in mtDNA content in human cancer. Int J Cancer 116, 920-4
Martinou, J.C. and Youle, R.J. (2011). Mitochondria in apoptosis: Bcl-2 family members and mitochondrial dynamics. Dev Cell 21, 92-101
Mehlen, P. and Puisieux, A. (2006). Metastasis: a question of life or death. Nat Rev Cancer 6, 449-58
Mizumachi, T. et al. (2008). Increased distributional variance of mitochondrial DNA content associated with prostate cancer cells as compared with normal prostate cells. Prostate 68, 408-17
Modica-Napolitano, J.S., Kulawiec, M. and Singh, K.K. (2007). Mitochondria and human cancer. Curr Mol Med 7, 121-31
Murphy, M.P. (2009). How mitochondria produce reactive oxygen species. Biochem J 417, 1-13
Paget, S. (1989). The distribution of secondary growths in cancer of the breast. 1889 [classical article]. Cancer & Metastasis Reviews 8, 98-101
Patanaphan, V., Salazar, O.M. and Risco, R. (1988). Breast cancer: metastatic patterns and their prognosis. South Med J 81, 1109-12
Scatena, R. (2012). Mitochondria and cancer: a growing role in apoptosis, cancer cell metabolism and dedifferentiation. Adv Exp Med Biol 942, 287-308
Schmidt-Kittler, O. et al. (2003). From latent disseminated cells to overt metastasis: genetic analysis of systemic breast cancer progression. Proc Natl Acad Sci U S A 100, 7737-42
Selvanayagam, P. and Rajaraman, S. (1996). Detection of mitochondrial genome depletion by a novel cDNA in renal cell carcinoma. Lab Invest 74, 592-9
Sotgia, F. et al. (2012). Mitochondrial metabolism in cancer metastasis: visualizing tumor cell mitochondria and the “reverse Warburg effect” in positive lymph node tissue. Cell Cycle 11, 1445-54
Steeg, P.S. (2006). Tumor metastasis: mechanistic insights and clinical challenges. Nat Med 12, 895-904
Taanman, J.W. (1999). The mitochondrial genome: structure, transcription, translation and replication. Biochim Biophys Acta 1410, 103-23
Tarassov, I., Kamenski, P., Kolesnikova, O., Karicheva, O., Martin, R.P., Krasheninnikov, I.A. and Entelis, N. (2007). Import of nuclear DNA-encoded RNAs into mitochondria and mitochondrial translation. Cell Cycle 6, 2473-7
Tseng, L.M., Yin, P.H., Chi, C.W., Hsu, C.Y., Wu, C.W., Lee, L.M., Wei, Y.H. and Lee, H.C. (2006). Mitochondrial DNA mutations and mitochondrial DNA depletion in breast cancer. Genes Chromosomes Cancer 45, 629-38
Weber, K., Ridderskamp, D., Alfert, M., Hoyer, S. and Wiesner, R.J. (2002). Cultivation in glucose-deprived medium stimulates mitochondrial biogenesis and oxidative metabolism in HepG2 hepatoma cells. Biol Chem 383, 283-90
Witkiewicz, A.K. et al. (2012). Using the "reverse Warburg effect" to identify high-risk breast cancer patients: stromal MCT4 predicts poor clinical outcome in triplenegative breast cancers. Cell Cycle 11, 1108-17
Yin, P.H. et al. (2004). Alteration of the copy number and deletion of mitochondrial DNA in human hepatocellular carcinoma. Br J Cancer 90, 2390-6
Yu, M. et al. (2007). Reduced mitochondrial DNA copy number is correlated with tumor progression and prognosis in Chinese breast cancer patients. IUBMB Life 59, 450-7
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, E.I. Mitochondrial dysfunction and cancer metastasis. J Bioenerg Biomembr 44, 619–622 (2012). https://doi.org/10.1007/s10863-012-9465-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10863-012-9465-9