Abstract
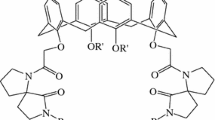
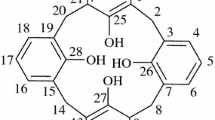
A novel two-step method was developed to prepare individual p-isopropenylcalix[n]arenes [n=4, 6, 8]. In the first step, linear phenolic oligomers were prepared in a basic medium from p-isopropenylphenol and paraformaldehyde. The second step, cyclization of the linear oligomers was carried out at higher temperatures. Ethylene glycol was used as medium, and sodium tetraborate-decahydrate as the catalyst. O-Acetylated derivatives were obtained by acetylation of the phenolic hydroxyl groups of p-isopropenylcalix[n]arenes [n=4, 6, 8]. The p-isopropenyl-calix[n]arenes and their acetylated derivatives were characterized by IR, 1H NMR and UV spectroscopy, and elemental analysis. Their ability to extract metal cations from aqueous solutions was evaluated via metal picrate extraction experiments. A parallel investigation of the cation-binding ability of the p-tert-butylcalix[n]arenes [n=4, 6, 8] and their acetylated derivatives was also performed. The p-isopropenylcalix[6]arene hexaacetate is the most effective extracting agent for metal picrates, and shows strong affinity towards Rb cation.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
For example: C.D. Gutsche: In J.F. Stoddart (ed.), Calixarenes Revisited, The Royal Society of Chemistry, Cambridge, UK (1998)
For example: (a) C.D. Gutsche and J.A. Levine: J. Am. Chem. Soc. 104, 2652 (1982). (b) C.D. Gutsche and P. F. Pagoria: J. Org. Chem. 50, 5795 (1985). (c) C.D. Gutsche and L. Lin: Tetrahedron 42, 1633 (1986). (d) C.D. Gutsche and K.C. Nam: J. Am. Chem. Soc. 110, 6153 (1988). (e) R. Lamartime, I. Dumazet, P.␣Choquard, A. Marcillac, F. Vocanson, and C. Duchamp: “Process for the preparation of calixarenes and new calixarene compounds”, U.S. Pat. No. 6,271,337 (2001)
(a) J. Vincens and V. Böhmer: In J.E.D. Davies (ed.), Calixarenes: A Versatile Class of Macrocyclic Compounds, Kluwer Academic Publishers, Dordrecht (1991). (b) Z. Asfari, V. Böhmer, J.␣Harrowfield, and J. Vicens (eds.), Calixarenes 2001, Kluwer Academic Publishers, Dordrecht (2001)
Mandolini L., Ungaro R. (eds.), (2000) Calixarenes in Action, Imperial College, London
(a) Reference 3, pp. 127–171, 211–215. (b) Reference 1, pp. 147–164. (c) R. Ludwig: Fresenius J. Anal. Chem. 367, 103 (2000)
Reference 1, pp. 169–177
For example see review: G. McMahon, S. O’Malley, K. Nolan, and D. Diamond: Arkivoc 23 (2003)
(a) F. Arnaud-Neu, E.M. Collins, M. Deasy, G. Ferguson, S.J.␣Harris, B. Kaitner, A.J. Lough, M.A. McKervey, M. Margues, B.L. Ruhl, M.J. Schwing-Weill, and E.M. Seward: J.␣Am. Chem. Soc. 111, 8681 (1989). (b) Reference 5, pp. 385–406
Casnati A., Pochini A., Ungaro R., Bocchi F., Ugozzoli F., Egberink R.J.M., Struijk H., Lugtenberg R., de Jong F., Reinhoudt D.N.: (1996). Chem. Eur. J. 2:436
Casnati A., Pochini A., Ungaro R., Bocchi F., Ugozzoli F., Arnaud F., Fanni S., Schwing M.J., Egberink R.J.M., de Jong F., Reinhoudt D.N.: (1995) J. Am. Chem. Soc. 117: 2767
(a) Reference 5, pp. 642–662. (b) F.J. Steemers, W. Verboom, D.N. Reinhoudt, E.B. van der Tol, and J.W. Verhoeven: J. Am. Chem. Soc. 117, 9408 (1995). (c) D.M. Rudkevich, W. Verboom, E.B. van der Tol, C.J. van Staveren, F.M. Kaspersen, J.W.␣Verhoeven, and D.N. Reinhoudt: J. Chem. Soc., Perkin Trans. 2 , 131 (1995)
Ikeda A., Shinkai S.: (1997). Chem. Rev. 97:1713
Y. Kubo: Synlett 161 (1999)
Molenveld P., Engbersen J.F.J., Kooijman H.J., Spek A.L., Reinhoudt D.N.: (1998) J. Am. Chem. Soc. 120:6726
T. Haino, H. Akii, and Y. Fukazawa: Synlett 1016 (1998)
Reference 3, p. 149
Reference 3, p. 173
Novakov P., Miloshev S., Tuleshkov P., Gitsov J., Georgieva M.: (1998). Die Angewandte Makromolekulare Chemie 255:23
Z.N. Verchovskaja: In I.V. Kalechica (ed.), Diphenilolpropan, Khimiya, Moskow (1971)
Gutsche C.D., Bauer L.J. (1985) J. Am. Chem. Soc. 107:6059
Wong K.H., Ng H.L.: (1981) J. Coord. Chem. 11:49
(a) L.D. Bratton, B. Strzelbicka, and R.A. Bartsch: Arkivoc 80 (2003). (b) Y.H. Cho, S.G. Rha, and S.-K. Chang: J. Incl. Phenom. Macrocyclic Chem. 31, 119 (1998). (c) G.M. Lein and D.J. Cram: J. Am. Chem. Soc. 107, 448 (1985)
For example: (a) R Ludwig: Fresenius J. Anal. Chem. 367, 103 (2000). (b) M. Mocerino, K. Mishima, H. Nishioka, A. Yoneda, and M. Ouchi: Anal. Sc. 1223 (2001)
For example: (a) R. Ludwig: JAERI, JAERI-Review 95-022, 152 (1995). (b) R. Ludwig and N. T. K. Dzung: Sensors 2, 397 (2002)
N. Mita: “Soluble calixarene derivative and films thereof”, U.S. Pat.No. 5,143,784 (1992)
Acknowledgements
The authors are indebted to the financial support from University of Chemical Technology and Metallurgy, Sofia, Bulgaria (Grant No 10143).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yordanova, S., Miloshev, S., Berlinova, I. et al. Synthesis and Cation-Binding Properties of Novel p-Isopropenylcalix[n]arenes and Their Acetylated Derivatives . J Incl Phenom Macrocycl Chem 56, 337–344 (2006). https://doi.org/10.1007/s10847-006-9104-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-006-9104-3