Abstract
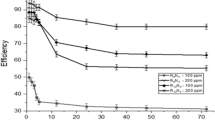
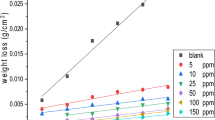
This work reports results of weight loss, potentiodynamic polarization and impedance measurements on the corrosion inhibition of copper in aerated non-stirred 3% NaCl solutions in the temperature range 15–65 °C using sodium oleate (SO) as an anionic surfactant inhibitor. These studies have shown that SO is a very good ”green”, mixed-type inhibitor. The inhibition process was attributed to the formation of an adsorbed film on the metal surface that protects the metal against corrosive agents. Scanning electron microscopy (SEM) and energy dispersion X-ray (EDX) observations of the electrode surface confirmed the existence of such an adsorbed film. The inhibition efficiency increases with increasing surfactant concentration and time of immersion, while it decreases with solution temperature. Maximum inhibition efficiency of the surfactant is observed at concentrations around its critical micellar concentration (CMC). The potential of zero charge (pzc) of copper was studied by ac impedance, and the mechanism of adsorption is discussed. The sigmoidal shape of the adsorption isotherm confirms the applicability of Frumkin’s equation to describe the adsorption process. Thermodynamic functions for the adsorption process were determined.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bacarella A.L., Griess J.C. (1973). J. Electrochem. Soc., 120: 459
W.H. Smyrl, in B.E. Conway, E. Yeager and R.E. White, (Eds), ‘Comprehensive Treatise of Electrochemistry’, Vol. 4, J.O’M. Bockris, (Plenum Press, New York, 1981), pp. 97–149.
Barcia O.E., Mattos O.R., Pebere N., Tribollet B. (1993). J. Electrochem. Soc. 140: 2825
Deslouis C., Tribollet B., Mengoli G., Musiani M.M. (1988). J. Appl. Electrochem. 18: 374
Benedetti A.V., Sumodjo P.T.A., Nobe K., Cabot P.L., Proud W.G. (1995). Electrochim. Acta 40: 2657
Zhou G., Shao H., Loo B.H. (1997) aa. J. Electroanal. Chem. 421: 129
Crundwell F.K. (1992). Electrochim. Acta 37: 2101
Lee H.P., Nobe K. (1986). J. Electrochem. Soc. 133: 2035
Fiaud C. (1995). 8th Eur. Symp. on Corrosion Inhibitors, Ann. Univ. Ferrara 2: 929
Gasparac R., Martin C.R., Stupnisek-Lisac E. (2000). J. Electrochem. Soc. 147: 548
Stupnisek-Lisac E., Gazivoda A., Madzarac M. (2002). Electrochim. Acta 47: 4189
Otmacic H., Stupnisek-Lisac E. (2003). Electrochim. Acta, 48: 985
F. Mansfeld and Y. Wang, in ‘Corrosion 95’, NACE, Paper No. 41, 1995.
Ma H., Chen S., Zhao S., Liu X., Liu D., Li D. (2001). J. Electrochem. Soc. 148: B482
G. Trabanelli in F. Mansfeld (Ed.), ‘Corrosion Mechanisms’, (Marcel Dekkar, Inc., New York, 1987), p. 119.
Luo H., Guan Y.C., Han K.N. (1998). Corrosion 54: 619
C.A. Miller and S. Qutubuddin, in H.-F, Eike and C.D. Parfitt (Eds), Interfacial Phenomena in polar Media, ’Surfactant Science Series’, Vol. 21, (Marcel Dekker, Inc., New York 1987), p. 166.
Ma H., Chen S., Yin B., Zhao S., Liu X. (2003). Corros. Sci. 45: 867
Abd El-Rehim S.S., Hassan H.H., Amin M.A. (2001). Mat. Chem. & Phys. 70: 64
Abd El-Rehim S.S., Hassan H.H., Amin M.A. (2002). Mat. Chem. & Phys. 78: 337
Abd El-Rehim S.S., Hassan H.H., Amin M.A. (2004). Corros. Sci. 46: 5–25
Tarasova N.S., Khachaturyan M.A. and Nikolaev L.A., (1984). Russ. J. Phys. Chem. 58:628
Boukamp B.A., (1990). Equivalent Circuit. Princeton Applied Research Corporation, Princeton, N J
Zhao T. and Mu G., (1999). Corros. Sci. 41:1937
M.J. Rosen, in ’Surfactants and Interfacial Phenomena’, (Wiley, New York, 1978), pp. 1–301.
Hassan H.H., (2001). Appl. Surf. Sci., 174:201
Bentiss F., Lagrence M., Traisnel M. and Hornez J.C., (1999). Corros. Sci. 41:789
Wu X., Ma H., Chen S., Xu Z. and Sui A., (1999). J. Electrochem. Soc. 146:1847
(a) B.A. Boukamamp, Solid State Ionics, 20 (1980) 31; (b) International Report CT 89/214/128, University of Twente, Eindhoven, The Netherlands (1989).
Moretti G., Guidi F. and Grion G., (2003). Corros. Sci. 46:387
J.O’M. Bockris, A.K.N. Reddy, in J.O’M. Bockris, B.E. Conway, E. Yeager and R.E. White (Eds), ’Modern Electrochemistry’, Vol. 2, (Plenum Press, New York, 1970), p. 708.
Damaskin B.B., Petrii O.A. and Batraktov B., (1971). Adsorption of Organic Compounds on Electrodes. Plenum Press, New York
Langmuir I., (1917). J. Am. Chem. Soc. 39:1848
Alberty R. and Silbey R., (1997). Physical Chemistry. Wiley, New York, p. 845
Bockris J.O’M. and Khan S.U.M., (1993). Surface Electrochemistry: A Molecular Level Approach. Plenum Press, New York
Schapinik J.W., Oudeman M., Leu K.W. and Helle J.N. (1960). Trans. Farad. Soc. 56:415
Frumkin A.N., (1925). Z. Phys. Chem. 116:466
Ikeda O., Jimbo H. and Tamura H., (1982). J. Electroanal. Chem. 137:127
J. Hill de Boer, ‘The Dynamical Character of Adsorption’, 2nd edn., (Clarendon Press, Oxford, UK, 1986).
Dhar H., Conway B. and Joshi K., (1973). Electrochim. Acta 18:789
Kamis E., Mellucci I., Lantanision R.M. and El-Ashry E.S.H. (1991). Corrosion 47:677
D. Do, ‘Adsorption Analysis: Equilibria and Kinetics’, (Imperial College Press, 1998), pp. 10–60.
Conway B., (1965). Principles of Electrode Processes. The Ronald Press Company, New York, pp. 78–85
Martinez S., (2003). Mat. Chem. & Phys. 77:97
Durnie W., De Marco R., Kinsella B. and Jefferson A., (1999). J. Electrochem. Soc. 146:1751
Martinez S. and Stern I., (2002). Appl. Surf. Sci. 199:83
Lampinen M.J. and Fomino M., (1993). J. Electrochem. Soc. 140:3537
Ishibashi M., Itoh M., Nishihara H. and Aramaki K., (1996). Electrochim. Acta 41:241
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Amin, M.A. Weight loss, polarization, electrochemical impedance spectroscopy, SEM and EDX studies of the corrosion inhibition of copper in aerated NaCl solutions. J Appl Electrochem 36, 215–226 (2006). https://doi.org/10.1007/s10800-005-9055-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10800-005-9055-1