Abstract
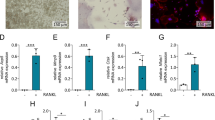
Chemokines are now known to play an important role in cancer growth and metastasis. Here we report that differentiating osteoclasts constitutively produce CCL22 (also called macrophage-derived chemokine) and potentially promote bone metastasis of lung cancer expressing its receptor CCR4. We first examined expression of chemokines by differentiating osteoclasts. CCL22 was selectively upregulated in osteoclast-like cells derived from RAW264.7 cells and mouse bone marrow cells upon stimulation with RANKL (receptor activator of nuclear factor-κB ligand). In addition, a human lung cancer cell line SBC-5 that efficiently metastasized to bone when intravenously injected into NK cell-depleted SCID mice was found to express CCR4. Stimulation of SBC-5 cells with CCL22 induced cell migration and also enhanced phosphorylation of protein kinase B/Akt and extracellular signal-regulated kinase (ERK). Furthermore, immunohistochemical analysis of bone metastasis lesions demonstrated close co-localization of tartrate-resistant alkaline phosphatase (TRAP)-positive osteoclasts expressing CCL22 and SBC-5 cells expressing CCR4. Collectively, these results suggest that osteoclasts may promote bone metastasis of cancer cells expressing CCR4 in the bone marrow by producing its ligand CCL22.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Fidler IJ (1973) Selection of successive tumour lines for metastasis. Nat New Biol 242:148–149
Nicolson GL (1988) Organ specificity of tumor metastasis: role of preferential adhesion, invasion and growth of malignant cells at specific secondary sites. Cancer Metastasis Rev 7:143–188
Nicolson GL (1988) Differential organ tissue adhesion, invasion, and growth properties of metastatic rat mammary adenocarcinoma cells. Breast Cancer Res Treat 12:167–176
Roodman GD (2004) Mechanisms of bone metastasis. N Engl J Med 350:1655–1664
Mundy GR (2002) Metastasis to bone: causes, consequences and therapeutic opportunities. Nat Rev Cancer 2:584–593
Yoneda T (2000) Cellular and molecular basis of preferential metastasis of breast cancer to bone. J Orthop Sci 5:75–81
Muller A, Homey B, Soto H et al. (2001) Involvement of chemokine receptors in breast cancer metastasis. Nature 410:50–56
Yoshie O, Imai T, Nomiyama H (1997) Novel lymphocyte-specific CC chemokines and their receptors. J Leukoc Biol 626:634–644
Taichman RS, Cooper C, Keller ET et al. (2002) Use of the stromal cell-derived factor-1/CXCR4 pathway in prostate cancer metastasis to bone. Cancer Res 62:1832–1837
Mochizuki H, Matsubara A, Teishima J et al. (2004) Interaction of ligand-receptor system between stromal-cell-derived factor-1 and CXC chemokine receptor 4 in human prostate cancer: a possible predictor of metastasis. Biochem Biophys Res Commun 320:656–663
Li H, Miyahara T, Tezuka Y et al. (1999) The effect of kampo formulae on bone resorption in vitro and in vivo. II. Detailed study of berberine. Biol Pharm Bull 22:391–396
Burstone MS (1958) Histochemical demonstration of acid phosphatases with naphthol AS-phosphates. J Natl Cancer Inst 21:523–539
Miki T, Yano S, Hanibuchi M et al. (2000) Bone metastasis model with multiorgan dissemination of human small-cell lung cancer (SBC-5) cells in natural killer cell-depleted SCID mice. Oncol Res 12:209–217
Nakamura ES, Koizumi K, Kobayashi M et al. (2004) Inhibition of lymphangiogenesis-related properties of murine lymphatic endothelial cells and lymph node metastasis of lung cancer by the matrix metalloproteinase inhibitor MMI270. Cancer Sci 95:25–31
Barlic J, McDermott DH, Merrell MN et al. (2004) Interleukin (IL)-15 and IL-2 reciprocally regulate expression of the chemokine receptor CX3CR1 through selective NFAT1- and NFAT2-dependent mechanisms. J Biol Chem 279:48,520–48,534
Lean JM, Murphy C, Fuller K et al. (2002) CCL9/MIP-1gamma and its receptor CCR1 are the major chemokine ligand/receptor species expressed by osteoclasts. J Cell Biochem 87:386–393
Okamatsu Y, Kim D, Battaglino R et al. (2004) MIP-1 gamma␣promotes receptor-activator-of-NF-kappa-B-ligand-induced osteoclast formation and survival. J Immunol 173:2084–2090
Mantovani A, Gray PA, Van Damme J et al. (2000) Macrophage-derived chemokine (MDC). J Leukoc Biol 68:400–404
Chan TO, Rittenhouse SE, Tsichlis PN (1999) AKT/PKB and other D3 phosphoinositide-regulated kinases: kinase activation by phosphoinositide-dependent phosphorylation. Annu Rev Biochem 68:965–1014
Zhou Y, Larsen PH, Hao C et al. (2002) CXCR4 is a major chemokine receptor on glioma cells and mediates their survival. J␣Biol Chem 277:49,481–49,487
Zhou H, Li XM, Meinkoth J et al. (2000) Akt regulates cell survival and apoptosis at a postmitochondrial level. J Cell Biol 151:483–94
Porcile C, Bajetto A, Barbero S et al. (2004) CXCR4 activation induces epidermal growth factor receptor transactivation in an ovarian cancer cell line. Ann N Y Acad Sci 1030:162–169
Scotton CJ, Wilson JL, Scott K et al. (2002) Multiple actions of the chemokine CXCL12 on epithelial tumor cells in human ovarian cancer. Cancer Res 62:5930–5938
Eisenhardt A, Frey U, Tack M et al. (2005) Expression analysis and potential functional role of the CXCR4 chemokine receptor in bladder cancer. Eur Urol 47:111–117
Li YM, Pan Y, Wei Y, Cheng X et al. (2004) Upregulation of CXCR4 is essential for HER2-mediated tumor metastasis. Cancer Cell 6:459–469
Benovic JL, Marchese A (2004) A new key in breast cancer metastasis. Cancer Cell 6:429–430
Murakami T, Cardones AR, Hwang ST (2004) Chemokine receptors and melanoma metastasis. J Dermatol Sci 36:71–78
Uchida D, Begum NM, Tomizuka Y et al. (2004) Acquisition of lymph node, but not distant metastatic potentials, by the overexpression of CXCR4 in human oral squamous cell carcinoma. Lab Invest 84:1538–1546
Acknowledgments
We thank Dr. Ryohei Ogawa (Department of Radiological Sciences, Faculty of Medicine) and Dr. Tatsuro Miyahara (Department of Toxicology, Faculty of Pharmaceutical Sciences) for helpful advice and Mr. Mamoru Nakamura and Dr. Hikaru Seto (Department of Radiology, Faculty of Medicine, Toyama Medical and Pharmaceutical University), Mr. Iwamoto Shinichi (Applied Medical Research Laboratory) for technical support. The authors also express their appreciation to Dr. Yoichi Kameda (Department of Pathology, Kanagawa Cancer Center), Dr. Kenya Shitara (Tokyo Research Laboratories, Kyowa Hakko Kogyo Co., Ltd), Dr. Kouji Matsushima (Department of Molecular Preventive Medicine, School of Medicine, The University of Tokyo) and Dr. Ryuzo Ueda (Department of Internal Medicine and Molecular Science, Nagoya City University Graduate School of Medical Science) for helpful suggestions.
Author information
Authors and Affiliations
Corresponding author
Additional information
This study was supported in part by a Grant-in-Aid for Young Scientists (B) (No. 15790089), Grant-in-Aids for Cancer Research (No. 16022224 and 16023225) and a Grant-in-Aid for the 21st Century COE Program from Ministry of Education, Culture, Sports, Science and Technology (MEXT), Japan, and by Solution Oriented Research for Science (SORST) of Japan Science and Technology Corporation (JST) and High-Tech Research Center Project for Private Universities: matching fund subsidy from MEXT, 2002–2006.
Rights and permissions
About this article
Cite this article
Nakamura, E.S., Koizumi, K., Kobayashi, M. et al. RANKL-induced CCL22/macrophage-derived chemokine produced from osteoclasts potentially promotes the bone metastasis of lung cancer expressing its receptor CCR4. Clin Exp Metastasis 23, 9–18 (2006). https://doi.org/10.1007/s10585-006-9006-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10585-006-9006-1