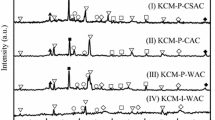
Various surface area (SBET) of cobaltic oxide (Co3O4) are prepared by different methods, i.e., precipitation–oxidation, impregnation and hydrothermal. The effect of SBET of Co3O4 on the catalytic property toward CO oxidation is investigated. The results indicate that the optimum SBET of Co3O4 could increase the catalytic activity. Characterization of the cobaltic oxide using X-ray diffraction (XRD), N2-adsorption at −196 °C, infrared (IR) and temperature-programmed reduction (TPR) reveals that the increase of SBET on Co3O4 can weaken the bond strength of Co–O and promote more lattice oxygen desorption from Co3O4 to cause the reduction become easy. We conclude that the SBET effect, caused by various prepared methods and refined conditions, are responsible for the activity enhancement of Co3O4. The T50 (the conversion of CO reached 50%) is decreased significantly when the SBET is increased, i.e., PO-R230 > PO-C400 > I-C550 > H-150 ~D-Strem.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Y. Kim S.K. Shi J.H. White (1980) J. Catal. 61 61
Y.Y. Yao (1984) J. Catal. 89 152
M. Olsbye R. Wendelbo T. Akporiayc (1997) Appl. Catal. A 152 127
F. Severino J. Laine (1983) Ind. Eng. Chem. Prod. Res. Dev. 22 396
W. Liu F.S. Maria (1995) J. Catal. 153 304
J. Jansson (2000) J. Catal. 194 55
J. Jansson A.E.C. Palmqvist E. Fridell M. Skoglundh L.O. Sterlund P. Thormahlen V. Langer (2002) J. Catal. 211 387
H.K. Lin C.B. Wang H.C. Chiu S.H. Chien (2003) Catal. Lett. 86 63
H.K. Lin H.C. Chiu H.C. Tsai S.H. Chien C.B. Wang (2003) Catal. Lett. 88 169
B.A. Sazonov V.V. Popovskii G.K. Boreskov (1968) Kinet. Catal. 9 255
D.S. Lafyatis G.P. Ansell S.C. Bennett J.C. Frost P.J. Millington R.R. Rajaram A.P. Walker T.H. Ballinger (1998) ArticleTitleApplied Catal. B 18 123
G.A. El-Shobaky N.M. Deraz (2001) Mater. Lett. 47 231
N.M. Deraz (2002) Colloids Surf. A 207 197
G.A. El-Shobaky A.M. Ghozza (2004) Mater. Lett. 58 699
H.P. Klug L.E. Alexander (1962) X-ray Diffraction Procedures for Polycrystalline and Amorphous Materials Wiley New York 491
C. Spenser D. Schroeder (1974) Phys. Rev. B 9 3658
T. Andrushkevich G. Boreskov V. Popovskii L. Pliasova L. Karakchiev A. Ostankovitch (1968) Kinet. Katal. 6 1244
G. Christoskova M. Stoyanova M. Georgieva D. Mehandjiev (1999) Mater. Chem. and Phys. 60 39
R.N. Singh J.P. Pandey N.K. Singh B. Lal P. Chartier J.F. Koenig (2000) Electrochim. Acta 45 1911
P. Arnoldy J.A. Moulijn (1985) J. Catal. 93 38
M. Voβ D. Borgmann G. Wedler (2002) J. Catal. 212 10
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Wang, CB., Tang, CW., Gau, SJ. et al. Effect of the surface area of cobaltic oxide on carbon monoxide oxidation. Catal Lett 101, 59–63 (2005). https://doi.org/10.1007/s10562-004-3750-0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10562-004-3750-0