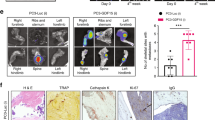
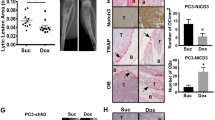
Abstract
Wnts are a large family of secreted glycoproteins that mediate bone development in the embryo and promote bone production in the adult. Autocrine Wnt signaling within tumor cells has been shown to promote tumorigenesis by enhancing tumor cell proliferation and survival. We recently demonstrated that prostate cancer cells (CaP) produce Wnts which act in a paracrine fashion to induce osteoblastic activity in CaP bone metastases. The ability of tumor-derived Wnts to influence bone development is regulated by multiple families of secreted antagonists including soluble frizzled related receptors (sFrp) and dickkopfs (DKK). CaP cells appear to produce DKK-1 early in the development of skeletal metastases, which masks osteogenic Wnts and thus favors an osteolytic environment at the metastatic site. As the metastases progresses, DKK-1 expression is lost allowing for a Wnt mediated osteoblastic response which predominates CaP boney lesions. Interestingly, blocking DKK-1 expression early in CaP metastasis prevents tumor establishment within the bone suggesting that osteolysis is a required first step in the development of CaP bone metastases. In this review, we discuss our data on the Wnt inhibitor DKK-1 in CaP bone metastasis in the context of current literature evidence that demonstrate that Wnt inhibitors can function as both tumor suppressors and tumor promoters. We provide a model that the affect of Wnt inhibitors on tumor development is dependent on the tumor micro-environment and suggest that DKK-1 is a switch which transitions CaP bone metastases from osteolytic to osteoblastic.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Logothetis, C. J., & Lin, S. H. (2005). Osteoblasts in prostate cancer metastasis to bone. Nature Reviews. Cancer, 5, 21–28.
Bubendorf, L., Schopfer, A., Wagner, U., Sauter, G., Moch, H., Willi, N., et al. (2000). Metastatic patterns of prostate cancer: An autopsy study of 1,589 patients. Human Pathology, 31, 578–583.
Roodman, G. D. (2004). Mechanisms of bone metastasis. New England Journal of Medicine, 350, 1655–1664.
Keller, E. T., Zhang, J., Cooper, C. R., Smith, P. C., McCauley, L. K., Pienta, K. J., et al. (2001). Prostate carcinoma skeletal metastases: Cross-talk between tumor and bone. Cancer Metastasis Reviews, 20, 333–349.
Logan, C. Y., & Nusse, R. (2004). The Wnt signaling pathway in development and disease. Annual Review of Cell and Developmental Biology, 20, 781–810.
Yanagawa, S., van Leeuwen, F., Wodarz, A., Klingensmith, J., & Nusse, R. (1995). The dishevelled protein is modified by wingless signaling in Drosophila. Genes & Development, 9, 1087–1097.
van de, W. M., Cavallo, R., Dooijes, D., van Beest, M, van Es, J., Loureiro, J., et al. (1997). Armadillo coactivates transcription driven by the product of the Drosophila segment polarity gene dTCF. Cell, 88, 789–799.
Kikuchi, A. (2000). Regulation of beta-catenin signaling in the Wnt pathway. [Review] [74 refs]. Biochemical & Biophysical Research Communications, 268, 243–248.
Yang, Y. (2003). Wnts and wing: Wnt signaling in vertebrate limb development and musculoskeletal morphogenesis. Birth Defects Research C Embryo Today Part C, Embryo Today, 69, 305–317.
Kuhl, M., Sheldahl, L.C., Park, M., Miller, J.R., Moon, R.T. (2000). The Wnt/Ca2+ pathway: A new vertebrate Wnt signaling pathway takes shape. [Review] [27 refs]. Trends in Genetics, 16, 279–283.
Veeman, M. T., Axelrod, J. D., & Moon, R. T. (2003). A second canon. Functions and mechanisms of beta-catenin-independent Wnt signaling. [Review] [94 refs]. Developments in Cell, 5, 367–377.
Bafico, A., Liu, G., Yaniv, A., Gazit, A., & Aaronson, S. A. (2001). Novel mechanism of Wnt signalling inhibition mediated by Dickkopf-1 interaction with LRP6/Arrow. Nature Cell Biology, 3, 683–686.
Mao, B., Wu, W., Li, Y., Hoppe, D., Stannek, P., Glinka, A., et al. (2001). LDL-receptor-related protein 6 is a receptor for Dickkopf proteins.[see comment]. Nature, 411, 321–325.
Mao, B., Wu, W., Davidson, G., Marhold, J., Li, M., Mechler, B. M., et al. (2002). Kremen proteins are Dickkopf receptors that regulate Wnt/beta-catenin signalling. Nature, 417, 664–667.
Buckwalter, J. A., Glimcher, M. J., Cooper, R. R., & Recker, R. (1996). Bone biology. I: Structure, blood supply, cells, matrix, and mineralization. Instructional Course Lectures, 45, 371–386.
Kostenuik, P. J. (2005). Osteoprotegerin and RANKL regulate bone resorption, density, geometry and strength. Current Opinion in Pharmacology, 5, 618–625.
Hall, C. L., Kang, S., Macdougald, O. A., & Keller, E. T. (2006). Role of wnts in prostate cancer bone metastases. Journal of Cellular Biochemistry, 97, 661–672.
Church, V. L., & Francis-West, P. (2002). Wnt signalling during limb development. Journal of Cellular Biochemistry, 46, 927–936.
Grotewold, L., & Ruther, U. (2002). The Wnt antagonist Dickkopf-1 is regulated by Bmp signaling and c-Jun and modulates programmed cell death. EMBO Journal, 21, 966–975.
Mukhopadhyay, M., Shtrom, S., Rodriguez-Esteban, C., Chen, L., Tsukui, T., Gomer, L., et al. (2001). Dickkopf1 is required for embryonic head induction and limb morphogenesis in the mouse. Developments in Cell, 1, 423–434.
Bodine, P. V., Zhao, W., Kharode, Y. P., Bex, F. J., Lambert, A. J., Goad, M. B., et al. (2004). The Wnt antagonist secreted frizzled-related protein-1 is a negative regulator of trabecular bone formation in adult mice. Molecular Endocrinology, 18, 1222–1237.
Giles, R. H., van Es, J. H., & Clevers, H. (2003). Caught up in a Wnt storm: Wnt signaling in cancer. Biochimica et Biophysica Acta, 1653, 1–24.
He, T. C., Sparks, A. B., Rago, C., Hermeking, H., Zawel, L., da Costa, L. T., et al. (1998). Identification of c-MYC as a target of the APC pathway.[comment]. Science, 281, 1509–1512.
Shtutman, M., Zhurinsky, J., Simcha, I., Albanese, C., D’Amico, M., Pestell, R., et al. (1999). The cyclin D1 gene is a target of the beta-catenin/LEF-1 pathway. Proceedings of the National Academy of Sciences of the United States of America, 96, 5522–5527.
Levy, L., Neuveut, C., Renard, C. A., Charneau, P., Branchereau, S., Gauthier, F., et al. (2002). Transcriptional activation of interleukin-8 by beta-catenin-Tcf4. Journal of Biological Chemistry, 277, 42386–42393.
Crawford, H. C., Fingleton, B. M., Rudolph-Owen, L. A., Goss, K. J., Rubinfeld, B., Polakis, P., et al. (1999). The metalloproteinase matrilysin is a target of beta-catenin transactivation in intestinal tumors. Oncogene, 18, 2883–2891.
Brabletz, T., Jung, A., Dag, S., Hlubek, F., & Kirchner, T. (1999). Beta-catenin regulates the expression of the matrix metalloproteinase-7 in human colorectal cancer. American Journal of Pathology, 155, 1033–1038.
Cronauer, M. V., Schulz, W. A., Ackermann, R., & Burchardt, M. (2005). Effects of WNT/beta-catenin pathway activation on signaling through T-cell factor and androgen receptor in prostate cancer cell lines. International Journal of Oncology, 26, 1033–1040.
Mulholland, D. J., Cheng, H., Reid, K., Rennie, P. S., & Nelson, C. C. (2002). The androgen receptor can promote beta-catenin nuclear translocation independently of adenomatous polyposis coli. Journal of Biological Chemistry, 277, 17933–17943.
Yardy, G. W., & Brewster, S. F. (2005). Wnt signalling and prostate cancer. Prostate Cancer and Prostatic Diseases, 8, 119–126.
Yang, X., Chen, M. W., Terry, S., Vacherot, F., Bemis, D. L., Capodice, J., et al. (2006). Complex regulation of human androgen receptor expression by Wnt signaling in prostate cancer cells. Oncogene, 25, 3436–3444.
Mulholland, D. J., Dedhar, S., Wu, H., & Nelson, C. C. (2006). PTEN and GSK3beta: Key regulators of progression to androgen-independent prostate cancer. Oncogene, 25, 329–337.
Sun, P., Xiong, H., Kim, T. H., Ren, B., & Zhang, Z. (2006). Positive inter-regulation between beta-catenin/T cell factor-4 signaling and endothelin-1 signaling potentiates proliferation and survival of prostate cancer cells. Molecular Pharmacology, 69, 520–531.
de la Taille, A., Rubin, M. A., Chen, M. W., Vacherot, F., de Medina, S. G., Burchardt, M., et al. (2003). Beta-catenin-related anomalies in apoptosis-resistant and hormone-refractory prostate cancer cells. Clinical Cancer Research, 9, 1801–1807.
Chen, G., Shukeir, N., Potti, A., Sircar, K., Aprikian, A., Goltzman, D., et al. (2004). Up-regulation of Wnt-1 and beta-catenin production in patients with advanced metastatic prostate carcinoma: Potential pathogenetic and prognostic implications. Cancer, 101, 1345–1356.
You, L., He, B., Xu, Z., Uematsu, K., Mazieres, J., Mikami, I., et al. (2004). Inhibition of Wnt-2-mediated signaling induces programmed cell death in non-small-cell lung cancer cells. Oncogene, 23, 6170–6174.
Weeraratna, A. T., Jiang, Y., Hostetter, G., Rosenblatt, K., Duray, P., Bittner, M., et al. (2002). Wnt5a signaling directly affects cell motility and invasion of metastatic melanoma. Cancer Cell, 1, 279–288.
Pukrop, T., Klemm, F., Hagemann, T., Gradl, D., Schulz, M., Siemes, S., et al. (2006). Wnt 5a signaling is critical for macrophage-induced invasion of breast cancer cell lines. Proceedings of the National Academy of Sciences of the United States of America, 103, 5454–5459.
Rubin, J. S., Barshishat-Kupper, M., Feroze-Merzoug, F., Xi, Z. F. (2006). Secreted WNT antagonists as tumor suppressors: Pro and con. Frontiers in Bioscience, 11, 2093–2105.
Aguilera, O., Fraga, M. F., Ballestar, E., Paz, M. F., Herranz, M., Espada, J., et al. (2006). Epigenetic inactivation of the Wnt antagonist DICKKOPF-1 (DKK-1) gene in human colorectal cancer. Oncogene, 25, 4116–4121.
Hall, C. L., Bafico, A., Dai, J., Aaronson, S. A., & Keller, E. T. (2005). Prostate cancer cells promote osteoblastic bone metastases through Wnts. Cancer Research, 65, 7554–7560.
Ugolini, F., Charafe-Jauffret, E., Bardou, V. J., Geneix, J., Adelaide, J., Labat-Moleur, F., et al. (2001). WNT pathway and mammary carcinogenesis: Loss of expression of candidate tumor suppressor gene SFRP1 in most invasive carcinomas except of the medullary type. Oncogene, 20, 5810–5817.
Wissmann, C., Wild, P. J., Kaiser, S., Roepcke, S., Stoehr, R., Woenckhaus, M., et al. (2003). WIF1, a component of the Wnt pathway, is down-regulated in prostate, breast, lung, and bladder cancer. Journal of Pathology, 201, 204–212.
Taniguchi, H., Yamamoto, H., Hirata, T., Miyamoto, N., Oki, M., Nosho, K., et al. (2005). Frequent epigenetic inactivation of Wnt inhibitory factor-1 in human gastrointestinal cancers. Oncogene, 24, 7946–7952.
Urakami, S., Shiina, H., Enokida, H., Kawakami, T., Tokizane, T., Ogishima, T., et al. (2006). Epigenetic inactivation of Wnt inhibitory factor-1 plays an important role in bladder cancer through aberrant canonical Wnt/beta-catenin signaling pathway. Clinical Cancer Research, 12, 383–391.
Mazieres, J., He, B., You, L., Xu, Z., Lee, A. Y., Mikami, I., et al. (2004). Wnt inhibitory factor-1 is silenced by promoter hypermethylation in human lung cancer. Cancer Research, 64, 4717–4720.
Zi, X., Guo, Y., Simoneau, A. R., Hope, C., Xie, J., Holcombe, R. F., et al. (2005). Expression of Frzb/secreted Frizzled-related protein 3, a secreted Wnt antagonist, in human androgen-independent prostate cancer PC-3 cells suppresses tumor growth and cellular invasiveness. Cancer Research, 65, 9762–9770.
Gonzalez-Sancho, J. M., Aguilera, O., Garcia, J. M., Pendas-Franco, N., Pena, C., Cal, S., et al. (2005). The Wnt antagonist DICKKOPF-1 gene is a downstream target of beta-catenin/TCF and is downregulated in human colon cancer. Oncogene, 24, 1098–1103.
Hsieh, S. Y., Hsieh, P. S., Chiu, C. T., & Chen, W. Y. (2004). Dickkopf-3/REIC functions as a suppressor gene of tumor growth. Oncogene, 23, 9183–9189.
Feng Han, Q., Zhao, W., Bentel, J., Shearwood, A. M., Zeps, N., Joseph, D., et al. (2006). Expression of sFRP-4 and beta-catenin in human colorectal carcinoma. Cancer Letter, 231, 129–137.
Wirths, O., Waha, A., Weggen, S., Schirmacher, P., Kuhne, T., Goodyer, C. G., et al. (2003). Overexpression of human Dickkopf-1, an antagonist of wingless/WNT signaling, in human hepatoblastomas and Wilms’ tumors. Laboratory Investigation, 83, 429–434.
Joesting, M. S., Perrin, S., Elenbaas, B., Fawell, S. E., Rubin, J. S., Franco, O. E., et al. (2005). Identification of SFRP1 as a candidate mediator of stromal-to-epithelial signaling in prostate cancer. Cancer Research, 65, 10423–10430.
Oshima, T., Abe, M., Asano, J., Hara, T., Kitazoe, K., Sekimoto, E., et al. (2005). Myeloma cells suppress bone formation by secreting a soluble Wnt inhibitor, sFRP-2. Blood, 106, 3160–3165.
Roth, W., Wild-Bode, C., Platten, M., Grimmel, C., Melkonyan, H. S., Dichgans, J., et al. (2000). Secreted Frizzled-related proteins inhibit motility and promote growth of human malignant glioma cells. Oncogene, 19, 4210–4220.
Tian, E., Zhan, F., Walker, R., Rasmussen, E., Ma, Y., Barlogie, B., et al. (2003). The role of the Wnt-signaling antagonist DKK1 in the development of osteolytic lesions in multiple myeloma.[see comment]. New England Journal of Medicine, 349, 2483–2494.
Glass, D. A., 2nd, Bialek, P., Ahn, J. D., Starbuck, M., Patel, M. S., Clevers, H., et al. (2005). Canonical Wnt signaling in differentiated osteoblasts controls osteoclast differentiation. Developments in Cell, 8, 751–764.
Zhang, J., Dai, J., Qi, Y., Lin, D.L., Smith, P., Strayhorn, C., et al. (2001). Osteoprotegerin inhibits prostate cancer-induced osteoclastogenesis and prevents prostate tumor growth in the bone. Journal of Clinical Investigation, 107, 1235–1244.
Gregory, C. A., Perry, A. S., Reyes, E., Conley, A., Gunn, W. G., & Prockop, D. J. (2005). Dkk-1-derived synthetic peptides and lithium chloride for the control and recovery of adult stem cells from bone marrow. Journal of Biological Chemistry, 280, 2309–2323.
Gunn, W. G., Conley, A., Deininger, L., Olson, S. D., Prockop, D. J., & Gregory, C. A. (2006). A crosstalk between myeloma cells and marrow stromal cells stimulates production of DKK1 and interleukin-6: A potential role in the development of lytic bone disease and tumor progression in multiple myeloma. Stem Cells, 24, 986–991.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hall, C.L., Keller, E.T. The role of Wnts in bone metastases. Cancer Metastasis Rev 25, 551–558 (2006). https://doi.org/10.1007/s10555-006-9022-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10555-006-9022-2